Online Database of Chemicals from Around the World
| Shouguang Fukang Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shouguangpharm.com | |||
 | +86 (536) 510-2364 510-3738 | |||
 | +86 (536) 510-7886 / 510-1568 | |||
 | info@shouguangpharm.com | |||
| Chemical manufacturer since 1993 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Wuxi Orient Detergent Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinobromide.com | |||
 | +86 (510) 8758-1290 | |||
 | +86 (510) 8758-3694 | |||
 | suguangming@126.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Qingdao Union Fine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.unionfinechem.com | |||
 | +86 (532) 6691-5948 | |||
 | +86 (532) 6691-6851 | |||
 | zsp@unionfinechem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Shandong Haoyuan Group | China | |||
|---|---|---|---|---|
 | www.haoyuangroup.cn | |||
 | +86 (536) 519-9628 | |||
 | +86 (536) 296-0566 | |||
 | fenginfo@gmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shouguang Nuomeng Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.nuomengchem.com | |||
 | +86 (536) 511-9508 | |||
 | +86 (536) 511-9508 | |||
 | nuomengchemholly@gmail.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Yancheng Longshen Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.longshenchem.com | |||
 | +86 (515) 8773-1896 +86 18261286666 | |||
 | +86 (515) 8865-2999 | |||
 | 2355756927@qq.com gaojunwyt@vip.163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Riverland Trading LLC. | USA | |||
|---|---|---|---|---|
 | www.riverlandtrading.com | |||
 | +1 (336).944-2293 | |||
 | bens@riverlandtrading.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2023 | ||||
| Anhui Weixiang New Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ahwxchem.com | |||
 | +86 13195623283 | |||
 | export@ahwxchem.com | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Hangzhou Hangsyn Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hangsyn.com | |||
 | +86 (571) 8546-3750 | |||
 | +86 (571) 8580-3035 | |||
 | sales@hangsyn.com | |||
| Chemical manufacturer | ||||
| Yancheng Creator Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.creatorchem.com | |||
 | +86 (515) 8871-0008 8871-0868 8307-2966 | |||
 | +86 (515) 8871-0858 / 8307-0966 | |||
 | yc@jssdchem.com | |||
| Chemical manufacturer | ||||
| ChemSampCo, Inc. | USA | |||
|---|---|---|---|---|
 | www.chemsampco.com | |||
 | +1 (609) 656-2440 | |||
 | +1 (609) 656-2446 | |||
 | sales@chemsampco.com | |||
| Chemical manufacturer since 1960 | ||||
| Yancheng Biaoye Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.biaoyechem.com | |||
 | +86 (515) 8856-3923 | |||
 | +86 (515) 8673-0988 | |||
 | cwb@biaoyechem.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Chemical pesticide >> Herbicide >> Amide herbicide |
|---|---|
| Name | 2-Bromopropane |
| Synonyms | Isopropyl bromide |
| Molecular Structure | 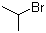 |
| Molecular Formula | C3H7Br |
| Molecular Weight | 122.99 |
| CAS Registry Number | 75-26-3 |
| EC Number | 200-855-1 |
| SMILES | CC(C)Br |
| Density | 1.3±0.1 g/cm3 Calc.*, 1.31 g/mL (Expl.) |
|---|---|
| Melting point | -89 °C (Expl.) |
| Boiling point | 60.6±8.0 °C 760 mmHg (Calc.)*, 59 °C (Expl.) |
| Flash point | 19.4 °C (Calc.)*, 19 °C (Expl.) |
| Solubility | water 0.3 g/100 mL (Expl.) |
| Index of refraction | 1.429 (Calc.)*, 1.425 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS02;GHS08 Danger Details GHS02;GHS08 Danger Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H225-H360F-H373 Details | ||||||||||||||||||||||||
| Safety Statements | P203-P210-P233-P240-P241-P242-P243-P260-P280-P303+P361+P353-P318-P319-P370+P378-P403+P235-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| Transport Information | UN 2344 | ||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
2-Bromopropane, also known as isopropyl bromide, is a simple secondary alkyl halide with the molecular formula C3H7Br. It is a colorless liquid with a mild ether-like odor and belongs to the class of haloalkanes, in which a bromine atom is covalently attached to the secondary carbon of a propyl group. Its secondary carbon-bromine bond makes it more reactive in nucleophilic substitution reactions than primary bromides in some contexts, while also being prone to elimination reactions under basic conditions. The discovery of 2-bromopropane stems from the development of bromination methods for alcohols in the 19th century. It was initially synthesized through the reaction of isopropanol with hydrobromic acid under acidic conditions, where the hydroxyl group of isopropanol is substituted with a bromine atom. Subsequent improvements included the use of phosphorus tribromide or other brominating agents to increase reaction efficiency and reduce byproducts. These methods established reliable routes for producing 2-bromopropane on both laboratory and industrial scales. 2-Bromopropane is widely used as an intermediate in organic synthesis. Its secondary bromide functionality allows it to act as an alkylating agent for a variety of nucleophiles. In pharmaceutical chemistry, it is employed to synthesize isopropylated derivatives of amines, alcohols, and heterocycles, which can influence biological activity, solubility, and metabolic stability. Its use in agrochemical production enables the construction of isopropyl-substituted intermediates for herbicides, insecticides, and fungicides. In polymer chemistry, 2-bromopropane can introduce isopropyl groups into monomers or polymer chains through nucleophilic substitution, creating branched or functionalized polymers. These modifications can enhance polymer flexibility, solubility, or reactivity for further chemical transformations. Its versatility also makes it useful in laboratory research as a reagent for mechanistic studies of nucleophilic substitution and elimination reactions, providing insights into reaction kinetics and stereochemical outcomes. Mechanistically, 2-bromopropane participates in both SN2 and SN1 reactions. The secondary carbon-bromine bond is more hindered than in primary bromides, which affects the reaction pathway depending on the nucleophile, solvent, and temperature. Strong nucleophiles in polar aprotic solvents typically favor SN2 substitution, while polar protic solvents and weak nucleophiles can lead to SN1 reactions with carbocation intermediates. Under strongly basic conditions, elimination reactions can produce propene. This dual reactivity makes 2-bromopropane a useful compound for studying organic reaction mechanisms. Physically, 2-bromopropane is slightly soluble in water but miscible with most organic solvents including ethers, alcohols, and hydrocarbons. It is highly flammable, volatile, and requires careful storage and handling in well-ventilated areas away from heat and ignition sources. Safety considerations are important, as 2-bromopropane can be toxic if inhaled or absorbed through the skin, and environmental exposure should be minimized. Proper protective equipment and disposal protocols are essential when working with this chemical. Overall, 2-bromopropane is an important secondary alkyl halide with diverse applications in organic synthesis, pharmaceuticals, agrochemicals, and polymer chemistry. Its reactivity as a bromide, versatility in substitution and elimination reactions, and utility in introducing isopropyl groups make it a valuable reagent for chemical research and industrial applications. References 2025. Novel 1,3,4-oxadiazole-nicotinamide hybrids as non-classical AHL mimics quorum sensing inhibitors of Pseudomonas aeruginosa: design, synthesis and biological evaluation. BMC Chemistry, 19. DOI: 10.1186/s13065-025-01560-9 2024. Ni-catalysed remote C(sp3)–H functionalization using chain-walking strategies. Nature Reviews Chemistry, 8(10). DOI: 10.1038/s41570-024-00649-4 2024. Repurposing of halogenated organic pollutants via alkyl bromide-catalysed transfer chlorination. Nature Chemistry, 16(6). DOI: 10.1038/s41557-024-01551-8 |
| Market Analysis Reports |