Online Database of Chemicals from Around the World
| Shandong Baoyuan Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.baoyuanchem.com | |||
 | +86 (533) 851-0343 628-8368 851-5988 | |||
 | +86 (533) 851-4528 / 628-3576 | |||
 | cnsdbaoyuan@163.com | |||
| Chemical manufacturer since 1983 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Zibo Xinghui Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xhhg.com | |||
 | +86 (533) 548-1773 | |||
 | +86 (533) 549-3977 | |||
 | xinghui@xhhg.com | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Chemtrade International Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.trade-chem.com | |||
 | +86 (532) 8689-3005 | |||
 | +86 (532) 8689-3005 | |||
 | chemtrade@trade-chem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Hubei Fuchi Chemical & Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuchigroup.com | |||
 | +86 (714) 753-1580 | |||
 | +86 (714) 753-1503 | |||
 | sales@fuchigroup.com | |||
| Chemical manufacturer since 1958 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Beckmann-Kenko GmbH | Germany | |||
|---|---|---|---|---|
 | www.beckmann-kenko.com | |||
 | +49 (4241) 930-888 | |||
 | +49 (4241) 930-889 | |||
 | info@beckmann-kenko.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| The Dow Chemical Company | USA | |||
|---|---|---|---|---|
 | www.dow.com | |||
 | +1 (989) 633-1706 | |||
 | fmdcigs@dow.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Cangzhou Enke Pharma-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.enkepharma.com | |||
 | +86 (317) 510-5699 510-6597 +86 15533709196 | |||
 | +86 (317) 510-6596 | |||
 | sale@enkepharma.com enkepharma@126.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: ymzhao | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Zhanhua Binbo Chemical | China | |||
|---|---|---|---|---|
 | www.binbochem.com | |||
 | +86 18853396269 | |||
 | 18853396269@139.com | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Riverland Trading LLC. | USA | |||
|---|---|---|---|---|
 | www.riverlandtrading.com | |||
 | +1 (336).944-2293 | |||
 | bens@riverlandtrading.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2023 | ||||
| Porton Corporation | China | |||
|---|---|---|---|---|
 | www.porton.cn | |||
 | +86 (23) 6763-5888 | |||
 | +86 (23) 6703-8513 / 6763-5889 | |||
 | porton@porton.cn | |||
| Chemical manufacturer since 2005 | ||||
| SelectLab Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.selectlab.de | |||
 | +49 (2383) 919-350 919-351 | |||
 | +49 (2383) 919-354 | |||
 | info@selectlab.de | |||
| Chemical manufacturer | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| AccuStandard Inc. | USA | |||
|---|---|---|---|---|
 | www.accustandard.com | |||
 | +1 (203) 786-5290 | |||
 | +1 (203) 786-5287 | |||
 | orders@accustandard.com | |||
| Chemical manufacturer since 1986 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Metal halides and halides >> Metal fluorides and salts |
|---|---|
| Name | Nitromethane |
| Synonyms | Nitrocarbol; NM |
| Molecular Structure | 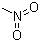 |
| Molecular Formula | CH3NO2 |
| Molecular Weight | 61.04 |
| CAS Registry Number | 75-52-5 |
| EC Number | 200-876-6 |
| SMILES | C[N+](=O)[O-] |
| Density | 1.1±0.1 g/cm3, Calc.*, 1.138 g/mL (Expl.) |
|---|---|
| Melting point | -29 °C (Expl.) |
| Index of Refraction | 1.358, Calc.*, 1.382 (Expl.) |
| Boiling Point | 85.1±3.0 °C (760 mmHg), Calc.*, 101.2 °C (Expl.) |
| Flash Point | 35.0 °C, Calc.*, 36 °C (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS02;GHS07;GHS08 Warning Details GHS02;GHS07;GHS08 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H302+H332-H302-H332-H351-H361d Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P210-P233-P240-P241-P242-P243-P261-P264-P270-P271-P280-P301+P317-P303+P361+P353-P304+P340-P317-P318-P330-P370+P378-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 1261 | ||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||
|
Nitromethane (CH3NO2) is an organic compound with a nitro group (-NO2) attached to a methyl group (-CH3). It is a colorless, oily liquid that is highly polar and has a slightly sweet odor. Nitromethane was first synthesized in the mid-19th century and has since found a variety of applications across different fields, including industrial, scientific, and military uses. The discovery of nitromethane dates back to the 19th century when it was first produced by nitrating methane using nitric acid. The compound’s structure was later elucidated, and its reactivity as an organic compound containing both a methyl and nitro group was explored. As the nitro group is an electron-withdrawing group, nitromethane exhibits distinct chemical properties compared to other simple hydrocarbons, which led to its use in various chemical processes. One of the primary uses of nitromethane is as a solvent and intermediate in chemical synthesis. It is used in the synthesis of various organic compounds, particularly those that are sensitive to moisture and require a non-aqueous environment for reactions. It is also utilized as a solvent in certain extraction processes and in the formulation of some pesticides and herbicides, where its polar nature and solvent properties are beneficial. Nitromethane is known for its use as a fuel in certain types of high-performance engines. It is commonly employed as a fuel additive in motorsport, particularly in drag racing. When combined with other fuels like methanol, nitromethane increases the power output of engines by providing more oxygen for combustion. This makes it a preferred fuel for some racing teams, as its energy density and combustion properties are ideal for performance applications. The compound’s high oxygen content facilitates more efficient combustion, producing more power and less soot. In the military, nitromethane is used in the production of explosives and propellants. Its ability to release a large amount of energy upon detonation makes it useful in the formulation of explosives. It is particularly important in the production of high-energy propellants for rocket engines. However, its use in this area is tightly regulated due to its potential for misuse and its association with explosives. In addition to its industrial and military applications, nitromethane has been investigated for use in pharmaceuticals and chemical research. It serves as an intermediate in the synthesis of certain drugs, where its ability to undergo specific chemical reactions is exploited. However, its use in drug production is limited due to its hazardous properties, and it is primarily used in laboratory settings for the synthesis of more complex compounds. Another important application of nitromethane is in its role as a reagent in the laboratory. It is used in the synthesis of nitroalkanes and as a source of the nitro group in reactions such as nucleophilic substitution and reductions. Its reactivity is utilized in the development of new chemical reactions and in the investigation of reaction mechanisms, particularly in organic chemistry. Nitromethane is also employed as a stabilizer in the production of certain types of plastics, particularly in the case of synthetic rubbers and other polymers. Its ability to stabilize reactive compounds is beneficial in manufacturing processes, where stability is crucial to avoid unwanted reactions during polymerization. In summary, nitromethane is an important chemical compound with a variety of applications. Its discovery in the 19th century opened the door to its use as a solvent, fuel, and intermediate in chemical synthesis. Today, it is primarily used in industrial applications, including as a fuel additive in motorsport, in the production of explosives and propellants, and as a reagent in chemical synthesis. Although it has a relatively narrow range of uses in pharmaceuticals, its versatility and unique properties continue to make it a valuable compound in various sectors of industry and research. References 2024. Oxidation of Hydrocarbons with H2O2/O2 Catalyzed by Osmium Complexes in Acetonitrile. Petroleum Chemistry, 64(6). DOI: 10.1134/s0965544124060227 2024. Visible Light Induced Eosin Y Catalysed Green Synthesis of Substituted Pyrroles. Catalysis Letters, 154(12). DOI: 10.1007/s10562-024-04888-4 2024. Calculation of Solute Partition Coefficient Using the A-P Scheme. Journal of Solution Chemistry, 53(10). DOI: 10.1007/s10953-024-01417-0 |
| Market Analysis Reports |