Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Isoquinoline compound |
|---|---|
| Name | 8-Bromo-1,2,3,4-tetrahydroisoquinoline |
| Molecular Structure | 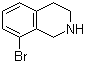 |
| Molecular Formula | C9H10BrN |
| Molecular Weight | 212.09 |
| CAS Registry Number | 75416-51-2 |
| EC Number | 817-034-4 |
| SMILES | C1CNCC2=C1C=CC=C2Br |
| Density | 1.4±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 294.3±40.0 °C 760 mmHg (Calc.)* |
| Flash point | 131.8±27.3 °C (Calc.)* |
| Index of refraction | 1.581 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
8-Bromo-1,2,3,4-tetrahydroisoquinoline is an organic compound with the molecular formula C9H10BrN. It belongs to the class of tetrahydroisoquinolines, which are bicyclic organic compounds containing a nitrogen atom in the ring structure. This particular derivative features a bromine atom attached at the 8-position of the tetrahydroisoquinoline ring. The discovery and study of tetrahydroisoquinoline derivatives have been of significant interest due to their potential biological activities. These compounds are often considered valuable scaffolds in medicinal chemistry and have been explored for their pharmacological properties, particularly in the context of their neuroactive effects. 8-Bromo-1,2,3,4-tetrahydroisoquinoline is particularly noteworthy in the synthesis of various bioactive molecules. The presence of the bromine atom at the 8-position enhances the molecule's reactivity and introduces potential sites for further functionalization. This can lead to the creation of more complex derivatives that could exhibit increased biological activity or improved pharmacokinetic properties. One of the primary applications of 8-bromo-1,2,3,4-tetrahydroisoquinoline is as a building block in the synthesis of other bioactive compounds, especially in the development of molecules targeting neurological conditions. Isoquinoline and its derivatives have been studied extensively for their potential as dopamine receptor agonists, antagonists, and inhibitors, which can be useful in the treatment of diseases like Parkinson's disease, schizophrenia, and addiction. The bromo group in the 8-position plays an important role in synthetic chemistry. It can serve as a point of substitution, allowing the introduction of various functional groups in order to tailor the molecule’s properties. This has been useful in developing compounds with selective activity against specific targets in the nervous system. The ability to modify the 8-bromo-1,2,3,4-tetrahydroisoquinoline structure offers flexibility in designing molecules that could interact with different types of receptors or enzymes, which is essential for drug discovery. Furthermore, tetrahydroisoquinoline derivatives, including 8-bromo-1,2,3,4-tetrahydroisoquinoline, have been studied for their potential anti-cancer properties. Some derivatives exhibit activity against certain types of cancer cells, making them a topic of interest for researchers exploring new chemotherapeutic agents. The bromine atom in this case could contribute to the compound’s ability to intercalate with DNA or disrupt other cellular processes, a common mechanism of action for various anticancer agents. In addition to its role in drug design, 8-bromo-1,2,3,4-tetrahydroisoquinoline is useful in the development of imaging agents. The compound’s ability to participate in chemical reactions involving metal ions, such as coordinating with transition metals, makes it a potential candidate for the creation of metal-based complexes that can be used in medical imaging or diagnostic applications. The synthetic pathways to 8-bromo-1,2,3,4-tetrahydroisoquinoline typically involve bromination reactions of 1,2,3,4-tetrahydroisoquinoline. This process introduces the bromine atom in a specific position of the aromatic ring, which can then be further functionalized to yield a variety of other useful derivatives. The synthesis of these compounds is an area of ongoing research, with methods continually being refined to improve yields, selectivity, and efficiency. As with many halogenated compounds, 8-bromo-1,2,3,4-tetrahydroisoquinoline must be handled with care in the laboratory, particularly because of the reactivity associated with the bromine atom. Proper safety protocols should be followed to minimize the risk of exposure to toxic fumes or other hazards during synthesis and handling. In conclusion, 8-bromo-1,2,3,4-tetrahydroisoquinoline is an important compound in organic synthesis, particularly in the development of bioactive molecules. Its use as a building block for the creation of complex molecules with potential neurological, anticancer, and imaging applications highlights its versatility. The presence of the bromine atom at the 8-position makes it a key starting material for further functionalization, and its study continues to provide valuable insights for drug discovery and medicinal chemistry. |
| Market Analysis Reports |