Online Database of Chemicals from Around the World
| Xi'an Tuochao Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xatuochao.cn | |||
 | +86 18092957403 | |||
 | chen.zeshan@xatuochao.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: +8618092957403 | |||
 | WhatsApp:+8618092957403 | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Classification | Chemical reagent >> Organic reagent >> Phosphonate / phosphonate |
|---|---|
| Name | Diethyl (2,2-diethoxyethyl)phosphonate |
| Synonyms | 2-diethoxyphosphoryl-1,1-diethoxyethane |
| Molecular Structure | 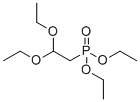 |
| Molecular Formula | C10H23O5P |
| Molecular Weight | 254.26 |
| CAS Registry Number | 7598-61-0 |
| EC Number | 231-504-0 |
| SMILES | CCOC(CP(=O)(OCC)OCC)OCC |
| Solubility | 5767 mg/L (25 °C water) |
|---|---|
| Density | 1.0±0.1 g/cm3, Calc.* |
| Index of Refraction | 1.424, Calc.* |
| Melting point | 50.85 °C |
| Boiling Point | 305.73 °C, 281.2±0.0 °C (760 mmHg), Calc.* |
| Flash Point | 155.5±45.4 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P271-P280-P302+P352-P305+P351+P338 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Diethyl (2,2-diethoxyethyl)phosphonate, commonly referred to as DEDEPh, is a chemical compound that has attracted attention for its unique properties and diverse applications in various fields, including agriculture, pharmaceuticals, and materials science. The discovery of this compound can be traced back to research efforts in the mid-20th century aimed at developing organophosphorus compounds with enhanced efficacy and reduced toxicity. Its synthesis typically involves the reaction of diethyl phosphite with 2,2-diethoxyethyl bromide, leading to the formation of this phosphonate ester. One of the primary applications of diethyl (2,2-diethoxyethyl)phosphonate is in the field of agricultural chemistry. It is utilized as an intermediate in the synthesis of phosphonate-based herbicides and insecticides. These compounds exhibit significant herbicidal and pesticidal activity, which makes them valuable for enhancing crop yields and protecting plants from pests and diseases. The phosphonate moiety in DEDEPh contributes to its biological activity by mimicking natural phosphorous compounds that plants and insects interact with. Research has shown that derivatives of diethyl (2,2-diethoxyethyl)phosphonate can be engineered to improve selectivity and efficacy, which is essential for developing sustainable agricultural practices. In addition to its agricultural applications, diethyl (2,2-diethoxyethyl)phosphonate has found utility in the pharmaceutical industry. It serves as a versatile building block for synthesizing various biologically active molecules. The ability to modify the phosphonate structure allows chemists to explore a range of potential drug candidates, particularly in the development of antiviral and anticancer agents. Research has demonstrated that compounds derived from diethyl (2,2-diethoxyethyl)phosphonate exhibit significant biological activity, making them candidates for further investigation in drug development. Furthermore, diethyl (2,2-diethoxyethyl)phosphonate is utilized in materials science, particularly in the development of advanced polymers and coatings. The phosphonate group can enhance the thermal stability and mechanical properties of materials, making them suitable for high-performance applications. For instance, polymers incorporating phosphonate functionalities are known to exhibit improved flame retardancy, which is crucial for various industrial applications. This characteristic has led to the exploration of diethyl (2,2-diethoxyethyl)phosphonate in the formulation of fire-resistant materials and coatings. The environmental implications of using diethyl (2,2-diethoxyethyl)phosphonate and its derivatives have been a subject of research as well. While phosphonates are generally considered less toxic than other organophosphorus compounds, studies are ongoing to assess their environmental fate and potential impact on ecosystems. The goal is to develop phosphonate-based products that minimize environmental risk while maximizing agricultural and industrial benefits. Safety is another critical aspect associated with diethyl (2,2-diethoxyethyl)phosphonate. As with many chemical substances, proper handling and storage protocols are essential to mitigate any potential health risks. Although DEDEPh is not classified as highly toxic, exposure to concentrated forms should be avoided to prevent irritation or other adverse effects. In summary, diethyl (2,2-diethoxyethyl)phosphonate is a versatile compound with significant applications in agriculture, pharmaceuticals, and materials science. Its discovery has facilitated advancements in these fields, particularly in the development of effective herbicides, insecticides, and drug candidates. Ongoing research into its properties and applications continues to reveal new potential uses, ensuring that diethyl (2,2-diethoxyethyl)phosphonate remains a valuable subject of study in both academic and industrial settings. References 1980. Cyclophosphamide potentiation and aldehyde oxidase inhibition by phosphorylated aldehydes and acetals. Journal of Medicinal Chemistry, 23(3). DOI: 10.1021/jm00177a018 2006. Condensation of resorcinol with phosphorylated acetals, synthesis of calix[4]resorcinolarenes with phosphorus-containing alkyl fragments in the lower rim. Russian Journal of General Chemistry, 76(3). DOI: 10.1134/s1070363206030108 2007. New phosphorus-containing analog of calix[4]resorcinarene based on 2,6-dihydroxypyridine. Russian Chemical Bulletin, 56(2). DOI: 10.1007/s11172-007-0060-x |
| Market Analysis Reports |