Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Wuhan Xinlaibokang Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xblkchem.com | |||
 | +86 18202741975 | |||
 | 3762793534@qq.com | |||
 | QQ Chat | |||
 | WeChat: 18202741975 | |||
| Chemical manufacturer since 2021 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Name | trans-Clomiphene citrate |
|---|---|
| Synonyms | (E)-Clomiphene citrate; Androxal; Clomiphene B citrate; Enclomid; Enclomiphene citrate |
| Molecular Structure | 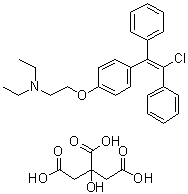 |
| Molecular Formula | C26H28ClNO.C6H8O7 |
| Molecular Weight | 598.08 |
| CAS Registry Number | 7599-79-3 |
| EC Number | 690-639-7 |
| SMILES | CCN(CC)CCOC1=CC=C(C=C1)/C(=C(C2=CC=CC=C2)/Cl)/C3=CC=CC=C3.C(C(=O)O)C(CC(=O)O)(C(=O)O)O |
| Solubility | 120 mg/mL (DMSO), <1 mg/mL (water) |
|---|---|
| Hazard Symbols |  GHS08 Danger Details GHS08 Danger Details |
|---|---|
| Risk Statements | H360-H361 Details |
| Safety Statements | P203-P280-P318-P405-P501 Details |
|
Trans-Clomiphene citrate, a stereoisomer of clomiphene citrate, is a significant compound in the field of reproductive medicine. Its discovery and application have had a profound impact on treating infertility and other reproductive health issues. Clomiphene citrate was first synthesized in the 1950s by chemists at Merrell Company, who were exploring new treatments for infertility. It was approved by the FDA in 1967. Clomiphene citrate consists of two geometric isomers: zuclomiphene (cis-clomiphene) and enclomiphene (trans-clomiphene). Trans-Clomiphene citrate, the more biologically active isomer, is responsible for the compound's therapeutic effects. Trans-Clomiphene citrate functions primarily as a selective estrogen receptor modulator (SERM). It binds to estrogen receptors in the hypothalamus, inhibiting the negative feedback of estrogen on gonadotropin release. This leads to an increase in the secretion of follicle-stimulating hormone (FSH) and luteinizing hormone (LH), which stimulate ovulation. As a result, trans-Clomiphene citrate is widely used in treating ovulatory dysfunction in women, particularly those with polycystic ovary syndrome (PCOS). The primary application of trans-Clomiphene citrate is in the treatment of female infertility. It is prescribed to induce ovulation in women who do not ovulate regularly or have difficulty conceiving due to hormonal imbalances. The treatment is typically administered orally, with the dosage and duration tailored to the patient’s specific needs. Beyond its use in female infertility, trans-Clomiphene citrate has also been explored for treating male infertility. By increasing the levels of FSH and LH, it can enhance spermatogenesis in men with certain types of hypogonadotropic hypogonadism. This expands its application and highlights its versatility as a therapeutic agent. Additionally, trans-Clomiphene citrate has been investigated for potential uses in treating other conditions related to hormonal imbalances, such as gynecomastia and secondary hypogonadism. Its ability to modulate estrogen receptors without completely blocking estrogen’s effects makes it a valuable tool in managing these conditions. The synthesis and production of trans-Clomiphene citrate involve a series of chemical reactions that isolate and purify the trans-isomer. Advances in chemical synthesis have improved the efficiency and purity of the compound, enhancing its therapeutic potential and reducing side effects. References 2014. Enclomiphene citrate stimulates testosterone production while preventing oligospermia: a randomized phase II clinical trial comparing topical testosterone. Fertility and Sterility, 102(3). DOI: 10.1016/j.fertnstert.2014.06.004 2016. Enclomiphene citrate for the treatment of secondary male hypogonadism. Expert Opinion on Pharmacotherapy, 17(11). DOI: 10.1080/14656566.2016.1204294 2023. Selective modulation of estrogen receptor in obese men with androgen deficiency: A systematic review and meta-analysis. Andrology, 11(1). DOI: 10.1111/andr.13373 |
| Market Analysis Reports |