Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hubei Seven Eight Nine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hb789.cn | |||
 | +86 (27) 8811-1833 +86 15802781837 | |||
 | info@hb789.cn niuxiudan@163.com | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Shanghai Fuqi Industrial & Trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fqsh.com | |||
 | +86 (21) 6631-0933 | |||
 | +86 (21) 6631-0933 | |||
 | fqchem@126.com | |||
| Chemical distributor since 2002 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Jiangsu Raien Environmental Protection Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.raien.cn | |||
 | +86 16651336278 | |||
 | 1780189359@qq.com | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Moto Chemistry Int'l Ltd. | China | |||
|---|---|---|---|---|
 | www.motochem.com | |||
 | +86 (756) 322-8461 322-8462 322-8463 | |||
 | +86 (756) 322-8460 | |||
 | moto@motochem.com | |||
| Chemical manufacturer | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| CABB AG | Switzerland | |||
|---|---|---|---|---|
 | www.cabb-chemicals.com | |||
 | +41 (61) 825-3111 | |||
 | +41 (61) 825-3636 | |||
 | contact@cabb-chemicals.com | |||
| Chemical manufacturer | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Metal sulfides and sulfates |
|---|---|
| Name | Sodium bisulfite |
| Synonyms | Monosodium sulfite; Sodium hydrogen sulfite |
| Molecular Structure | 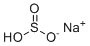 |
| Molecular Formula | NaHSO3 |
| Molecular Weight | 104.05 |
| CAS Registry Number | 7631-90-5 |
| EC Number | 231-548-0 |
| SMILES | OS(=O)[O-].[Na+] |
| Density | 1.48 |
|---|---|
| Melting point | 150 °C |
| Water solubility | 300 g/L |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302 Details | ||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P264-P270-P301+P317-P330-P501 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2693 | ||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
Sodium bisulfite (NaHSO₃) was first prepared in the early 19th century, when chemists began to explore the properties of sulfur dioxide (SO₂) and its reactions with alkaline substances. Although there are few exact records of its initial synthesis, this compound emerged from extensive research on sulfur-containing chemicals, primarily discovered by researchers such as French chemist Claude Louis Berthollet who studied sulfur dioxide and its salts. Sodium bisulfite has several unique properties. It appears as a white crystalline solid or powder, has a molecular weight of 104.06 g/mol, decomposes before melting, and is soluble in water to form weak acidic solutions. NaHSO₃ is known for its reducing properties and ability to release sulfur dioxide in aqueous solutions, which can be used in a variety of chemical processes. Sodium bisulfite is commonly used in water treatment to effectively neutralize chlorine and chloramines in water. This application is critical for municipal water treatment facilities and industries that need to remove chlorine to prevent corrosion or damage to equipment. In the chemical industry, sodium bisulfite acts as a reducing agent in a variety of chemical reactions. For example, it is used to reduce aldehydes and ketones in organic synthesis, helping to produce different compounds. It provides controlled release of sulfur dioxide in processes where it is needed, such as in the manufacture of certain dyes and fruit preservation. Sodium bisulfite is used in food processing as a preservative and antioxidant for dried fruits, wines, and other foods. It helps prevent spoilage and discoloration by inhibiting the growth and oxidation of microorganisms. In the textile industry, sodium bisulfite is used as a bleaching agent for various fibers and textiles. Its reducing properties help remove colorants and stains from fabrics, enhancing the appearance of fabrics. Sodium bisulfite is also used in photography and is an ingredient in black and white photographic developer solutions. It helps control pH and reduces oxidants, which is essential for proper film development. Handling sodium bisulfite requires safety precautions, use gloves, goggles, and lab coats to prevent contact with skin and eyes. Work in a well-ventilated area or use a fume hood to avoid breathing dust or vapors. Store in a cool, dry place in sealed containers to prevent moisture absorption, which can cause the release of sulfur dioxide. The use of sodium bisulfite must consider environmental and health effects and should be handled properly to avoid environmental contamination. Its use in water treatment should be managed to prevent the release of excessive sulfur dioxide into natural water sources. Exposure can cause irritation of the skin, eyes, and respiratory system. Proper safety measures are essential to minimize health risks. References 2003. 3-Carboxy-4-phosphonocyclopentane amino acids: New metabotropic glutamate receptor ligands. Amino Acids, 24(3). DOI: 10.1007/s00726-002-0405-6 2012. Biological denitrification of brine: the effect of compatible solutes on enzyme activities and fatty acid degradation. Biodegradation, 23(5). DOI: 10.1007/s10532-012-9542-0 2024. The synthesis of indane derivatives and antioxidant effects. Monatshefte für Chemie - Chemical Monthly, 155(11). DOI: 10.1007/s00706-024-03267-4 |
| Market Analysis Reports |