Online Database of Chemicals from Around the World
| Fujian South Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.southpharma.com | |||
 | +86 (598) 286-0412 +86 13509335186 | |||
 | +86 (598) 286-0416 | |||
 | sales@southpharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Beijing Mesochem Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.mesochem.com | |||
 | +86 (10) 5786-2036 57862181 67374028 +86 13366977697 | |||
 | +86 (10) 5786-2181 | |||
 | sales@mesochem.com huafenginfo@126.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Biochempartner Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.biochempartner.com | |||
 | +86 (27) 5923-9227 | |||
 | +86 (27) 5923-9227 | |||
 | 2323744551@qq.com | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shandong Boyuan Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.boyuanpharm.com | |||
 | +86 (531) 6995-4981 8896-3280 +86 15806417970 | |||
 | +86 (531) 8896-4879 | |||
 | Jeffrey.Liu@boyuanpharm.com boyuanchem@126.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Aktin Chemicals Inc | China | |||
|---|---|---|---|---|
 | www.aktinchem.com | |||
 | +86 400-028-7725 | |||
 | +86 (28) 8515-2372 | |||
 | info@aktinchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Alfa Chemistry | USA | |||
|---|---|---|---|---|
 | www.alfa-chemistry.com | |||
 | +1 (201) 478-8534 | |||
 | +1 (516) 927-0118 | |||
 | inquiry@alfa-chemistry.com | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Qichuang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qc-chemical.com | |||
 | +86 (571) 8893-5129 | |||
 | +86 (571) 8825-0182 | |||
 | david@qc-chemical.com sales@qc-chemical.com davidw0828@gmail.com | |||
 | QQ Chat | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Selleck Chemicals LLC | USA | |||
|---|---|---|---|---|
 | www.selleckchem.com | |||
 | +1 (713) 535-9129 | |||
 | +1 (832) 582-8590 | |||
 | info@selleckchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Smartchem (Beijing) Ltd. | China | |||
|---|---|---|---|---|
 | www.smartchembeijing.com | |||
 | +86 (10) 5720-3829 5130-5129 +86 13301124250 | |||
 | +86 (10) 5130-5388 | |||
 | sales@smartchembeijing.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Renheng Pharm Limited | China | |||
|---|---|---|---|---|
 | www.renheng.com.cn | |||
 | +86 (20) 3998-1653 | |||
 | +86 (20) 3998-1158 | |||
 | sales@renheng.com.cn | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Cangzhou Enke Pharma-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.enkepharma.com | |||
 | +86 (317) 510-5699 510-6597 +86 15533709196 | |||
 | +86 (317) 510-6596 | |||
 | sale@enkepharma.com enkepharma@126.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: ymzhao | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shenzhen Tianyuan Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tianpharm.com | |||
 | +86 (755) 8526-9922 | |||
 | sales3@tianpharm.com | |||
| Chemical manufacturer since 2018 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Anqing World Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldchem.cn | |||
 | +86 18155670904 | |||
 | flint@worldpharma.cn | |||
 | QQ Chat | |||
| Chemical distributor since 2010 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Targetmol Chemicals Inc. | USA | |||
|---|---|---|---|---|
 | www.targetmol.com | |||
 | +1 (781) 999-5354 | |||
 | +1 (781) 281-9145 | |||
 | sales@targetmol.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Exclusive Chemistry Ltd. | Russia | |||
|---|---|---|---|---|
 | www.exchemistry.com | |||
 | +7 (903) 713-5556 | |||
 | +7 (484) 396-4302 | |||
 | info@exchemistry.com | |||
| Chemical manufacturer | ||||
| Axon MedChem BV | Netherlands | |||
|---|---|---|---|---|
 | www.axonmedchem.com | |||
 | +31 (50) 311-8007 | |||
 | +31 (50) 360-0390 | |||
 | order@axonmedchem.com | |||
| Chemical manufacturer | ||||
| W & J PharmaChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.wjpharmachem.com | |||
 | +1 (301) 880-0624 | |||
 | +1 (301) 434-9366 | |||
 | info@wjpharmachem.com | |||
| Chemical manufacturer | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Inhibitor >> DNA damage >> PARP inhibitor |
|---|---|
| Name | Olaparib |
| Synonyms | 1-(Cyclopropylcarbonyl)-4-[5-[(3,4-dihydro-4-oxo-1-phthalazinyl)methyl]-2-fluorobenzoyl]piperazine |
| Molecular Structure | 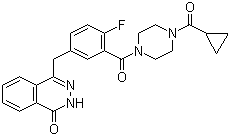 |
| Molecular Formula | C24H23FN4O3 |
| Molecular Weight | 434.46 |
| CAS Registry Number | 763113-22-0 (937799-91-2) |
| EC Number | 642-941-5 |
| SMILES | C1CC1C(=O)N2CCN(CC2)C(=O)C3=C(C=CC(=C3)CC4=NNC(=O)C5=CC=CC=C54)F |
| Density | 1.4±0.1 g/cm3 Calc.* |
|---|---|
| Solubility | DMSO 81mg/mL, Water $lessThan$1.2mg/mL, Ethanol $lessThan$1.2mg/mL (Expl.) |
| Index of refraction | 1.702 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS06;GHS08;GHS09 Danger Details GHS06;GHS08;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H341-H360-H360D-H372-H411 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P260-P264-P270-P273-P280-P301+P316-P318-P319-P321-P330-P391-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
Olaparib is a medication that belongs to a class of drugs known as PARP inhibitors. It was developed by AstraZeneca and is primarily used in the treatment of certain types of cancers, particularly those with specific genetic mutations. Olaparib works by inhibiting the enzyme poly(ADP-ribose) polymerase (PARP), which plays a key role in DNA repair processes within cells. The discovery of olaparib was rooted in the understanding of DNA damage repair mechanisms and how cancers, particularly those associated with BRCA1 and BRCA2 gene mutations, could be treated by targeting these repair processes. It was found that cancer cells, especially those with defects in the BRCA genes, rely heavily on other repair pathways, such as those involving PARP, to fix DNA damage and continue to grow. By inhibiting PARP, olaparib prevents the repair of DNA damage in these cancer cells, leading to cell death. This approach is referred to as "synthetic lethality," where the inhibition of two pathways (in this case, BRCA defects and PARP inhibition) leads to cell death, but normal cells, which have functional BRCA genes, are less affected. Olaparib was first approved for use in the treatment of ovarian cancer in patients with a BRCA mutation. Since then, its indications have expanded to include other cancers, including breast cancer, prostate cancer, and pancreatic cancer, particularly those with BRCA mutations or other genetic defects that affect DNA repair mechanisms. In these cancers, olaparib is often used as part of a targeted therapy regimen, either alone or in combination with other treatments such as chemotherapy or other forms of immunotherapy. The use of olaparib has shown significant efficacy in improving progression-free survival in patients with advanced cancers, especially those with BRCA mutations. In clinical trials, olaparib has demonstrated the ability to delay disease progression and improve outcomes compared to standard treatments. This has made it a valuable option for patients with cancers that are resistant to traditional therapies. Additionally, the drug is typically administered orally, providing a convenient and less invasive treatment option for patients. Olaparib’s role in cancer therapy is particularly important because it represents a shift towards personalized medicine. By targeting specific genetic mutations, olaparib provides an approach that is more tailored to the individual patient’s cancer profile. This precision medicine approach is expected to lead to more effective treatments with fewer side effects compared to traditional chemotherapies, which often affect both cancerous and healthy cells. In addition to its use in cancer, olaparib is also being investigated in clinical trials for other potential applications. These include studies looking at its effectiveness in combination with other cancer therapies, its use in cancers with different genetic mutations, and its potential for use in treating rare genetic disorders that also involve DNA repair deficiencies. However, as with any medication, olaparib is not without side effects. The most common adverse effects include nausea, fatigue, anemia, and various gastrointestinal issues. More serious side effects can include bone marrow suppression and an increased risk of developing other cancers. As a result, patients receiving olaparib must be carefully monitored, and its use is typically restricted to those with specific genetic profiles. In conclusion, olaparib is a significant advancement in cancer treatment, particularly for patients with cancers that have genetic mutations affecting DNA repair. Its ability to exploit synthetic lethality to kill cancer cells with defective DNA repair mechanisms makes it a promising therapy, particularly in the context of personalized medicine. Ongoing research into olaparib’s applications in combination with other therapies continues to expand its potential, making it an important tool in the fight against cancer. References 2014. PARP Inhibition Restores Extrinsic Apoptotic Sensitivity in Glioblastoma. PLoS ONE, 9(12). DOI: 10.1371/journal.pone.0114583 2018. Long-term efficacy, tolerability and overall survival in patients with platinum-sensitive, recurrent high-grade serous ovarian cancer treated with maintenance olaparib capsules following response to chemotherapy. British Journal of Cancer, 119(9). DOI: 10.1038/s41416-018-0271-y 2018. Maintenance Olaparib in Patients with Newly Diagnosed Advanced Ovarian Cancer. The New England Journal of Medicine, 379(26). DOI: 10.1056/nejmoa1810858 |
| Market Analysis Reports |