Online Database of Chemicals from Around the World
| Jinan Chenghui-Shuangda Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jnchsd.com | |||
 | +86 (531) 5889-7051 +86 15053146086 | |||
 | +86 (531) 5889-7093 | |||
 | jnchsd@qq.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Nanjing Legend Pharmaceutical & Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.legendpharm.com | |||
 | +86 (25) 8317-2486 | |||
 | +86 (25) 8317-2489 | |||
 | sales@legendpharm.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Changzhou Hi-Tech Chemistry Corp | China | |||
|---|---|---|---|---|
 | www.eastfine.net | |||
 | +86 (519) 8662-7600 | |||
 | +86 (519) 8662-9716 | |||
 | sales@watsonchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Shandong Chengchuang Pharmaceutical Technology Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ccmed.cn | |||
 | +86 (531) 5889-7091 | |||
 | +86 (531) 5889-7092 | |||
 | ccyychem@163.com | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hebei Brant Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.brantpharm.com | |||
 | +86 18713118368 | |||
 | lisa@brantpharm.com | |||
 | WhatsApp:0086 18713118368 | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Sinbond Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinbondpharm.com | |||
 | +86 (531) 8703-8285 +86 13583181986 +44 (208) 242-5518 | |||
 | +86 (531) 8703-8285 | |||
 | cici1124@gmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| Classification | Flavors and spices >> Synthetic spice >> Lactone and oxygen-containing heterocyclic compound >> Thiazole, thiophene and pyridine |
|---|---|
| Name | 4-(4-Pyridinyl)thiazole-2-thiol |
| Synonyms | 4-pyridin-4-yl-3H-1,3-thiazole-2-thione |
| Molecular Structure | 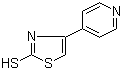 |
| Molecular Formula | C8H6N2S2 |
| Molecular Weight | 194.28 |
| CAS Registry Number | 77168-63-9 |
| EC Number | 616-441-2 |
| SMILES | C1=CN=CC=C1C2=CSC(=S)N2 |
| Density | 1.4$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 356.1$+/-$34.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 169.2$+/-$25.7 $degree$C (Calc.)* |
| Index of refraction | 1.671 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H317-H413 Details | ||||||||||||||||
| Safety Statements | P261-P264-P270-P272-P273-P280-P301+P317-P302+P352-P321-P330-P333+P317-P362+P364-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
4-(4-Pyridinyl)thiazole-2-thiol is a heterocyclic aromatic compound featuring a thiazole ring substituted at the 4-position with a pyridinyl group and a thiol group at the 2-position. Compounds of this type are significant in medicinal chemistry, agrochemicals, and coordination chemistry due to the combination of sulfur- and nitrogen-containing heterocycles, which provide nucleophilic, hydrogen-bonding, and metal-coordinating sites. The thiol group is a reactive nucleophile capable of forming disulfides, thioethers, or metal complexes, while the pyridine ring introduces additional electron density and planarity for interactions with biological targets or synthetic elaboration. Structurally, the molecule consists of a five-membered thiazole ring with a sulfur atom at position 1, a nitrogen atom at position 3, a thiol group at position 2, and a pyridine ring connected at position 4. The thiazole ring is aromatic, contributing to planarity and conjugation with the attached pyridine ring. The thiol group is acidic and nucleophilic, allowing selective reactions with electrophiles, while the pyridinyl moiety can participate in hydrogen bonding, metal coordination, or further functionalization. This combination of heterocycles and functional groups makes the molecule versatile in both synthetic and bioactive contexts. The synthesis of 4-(4-pyridinyl)thiazole-2-thiol typically involves condensation reactions between α-haloketones or α-haloesters and thiourea derivatives to form the thiazole ring, followed by palladium-catalyzed cross-coupling or nucleophilic substitution to attach the pyridinyl group at the 4-position. Reaction conditions are carefully controlled to maintain the integrity of the thiol group and the heterocyclic framework. Purification yields a stable, crystalline product suitable for further derivatization. In medicinal chemistry, thiazole-thiol derivatives are investigated as enzyme inhibitors, receptor ligands, and metal-binding agents. The thiol group can interact with cysteine residues in proteins or form reversible covalent bonds with reactive centers, while the pyridinyl ring enhances binding affinity and selectivity through π–π stacking or hydrogen-bonding interactions. The aromatic thiazole provides rigidity and planarity, which are beneficial for receptor recognition or enzyme active site complementarity. This structure allows for the design of bioactive molecules with tunable pharmacokinetic and pharmacodynamic properties. Beyond pharmaceutical applications, 4-(4-pyridinyl)thiazole-2-thiol is a valuable intermediate in organic synthesis and coordination chemistry. The thiol group enables the formation of thioethers, disulfides, and metal complexes, while the heterocyclic rings can undergo further functionalization through halogenation, alkylation, or coupling reactions. This versatility allows chemists to construct heterocyclic libraries, functionalized materials, and organometallic complexes for catalysis or chemical biology applications. Overall, 4-(4-pyridinyl)thiazole-2-thiol is a multifunctional heterocyclic compound combining a thiazole ring, a thiol group, and a pyridinyl substituent. Its nucleophilic, aromatic, and hydrogen-bonding properties make it a versatile intermediate for organic synthesis, medicinal chemistry, and the development of bioactive compounds and functional materials. References 2017. Synthesis of 4- and 5-arylthiazolinethiones as inhibitors of indoleamine 2,3-dioxygenase. Bioorganic & Medicinal Chemistry Letters. DOI: 10.1016/j.bmcl.2016.06.052 S109 | PARCEDC | List of 7074 potential endocrine disrupting compounds (EDCs) by PARC T4.2. DOI: 10.5281/zenodo.10944198 |
| Market Analysis Reports |