Online Database of Chemicals from Around the World
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Shanghai Sinofluoro chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinofluoro.com | |||
 | +86 (21) 6427-9360 | |||
 | +86 (21) 6427-8603 | |||
 | simon.lv@sinofluoro.com jerry.chen@sinofluoro.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Suzhou Ryan Pharmachem Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ryanchem.com | |||
 | +86 (512) 6878-0025 6878-0026 +86 18013195319 | |||
 | +86 (512) 6878-0025 ex 8004 | |||
 | sales@ryanchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Changzhou Boren Chem-Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.borenchem.cn | |||
 | +86 (519) 8208-5178 +86 13328197671 | |||
 | +86 (519) 8585-9553 | |||
 | sales@borenchem.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Coresyn Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.coresyn.com | |||
 | +86 (571) 8662-6709 | |||
 | sales@coresyn.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai CR Corporation Limited | China | |||
|---|---|---|---|---|
 | www.crcorporation.cn | |||
 | +86 (21) 5736-6208 +86 13917420128 | |||
 | +86 (21) 5736-6228 | |||
 | fred.wen@crcorporation.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Changzhou Jiuwu Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jiuwuchem.com | |||
 | +86 (519) 8512-1086 +86 13775162768 | |||
 | +86 (519) 8512-5369 | |||
 | info@jiuwuchem.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Combi-Blocks, Inc. | USA | |||
|---|---|---|---|---|
 | www.combi-blocks.com | |||
 | +1 (858) 635-8950 | |||
 | +1 (858) 635-8991 | |||
 | sales@combi-blocks.com | |||
| Chemical manufacturer | ||||
| Dhruvtara Chemicals Pvt Ltd. | India | |||
|---|---|---|---|---|
 | www.dhruvtarachemicals.com | |||
 | +91 (40) 2715-4607 | |||
 | +91 (40) 2715-4607 | |||
 | info@dhruvtarachemicals.com | |||
| Chemical manufacturer | ||||
| Atlantic Research Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.atlantic-chemicals.com | |||
 | +44 (8707) 746-454 | |||
 | +44 (8707) 746-455 | |||
 | info@atlantic-chemicals.com | |||
| Chemical manufacturer | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| GLSynthesis Inc. | USA | |||
|---|---|---|---|---|
 | www.glsynthesis.com | |||
 | +1 (508) 754-6700 | |||
 | +1 (508) 754-7075 | |||
 | support@glsynthesis.com | |||
| Chemical manufacturer | ||||
| HetCat - Heterocycles & Catalysts | Switzerland | |||
|---|---|---|---|---|
 | www.hetcat.com | |||
 | +41 (61) 363-9868 | |||
 | +41 (61) 363-9869 | |||
 | fallahpour@hetcat.com | |||
| Chemical manufacturer | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Carbone Scientific Co., Ltd. | UK | |||
|---|---|---|---|---|
 | www.carbonesci.com | |||
 | +44 (870) 486-8629 | |||
 | +44 (870) 288-7399 | |||
 | sales@carbonesci.com | |||
| Chemical distributor | ||||
| AOBChem USA | USA | |||
|---|---|---|---|---|
 | www.aobchem.com | |||
 | +1 (323) 382-7678 | |||
 | +1 (213) 687-8698 | |||
 | sales@aobchem.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Anisyn, Inc. | USA | |||
|---|---|---|---|---|
 | www.anisyn.com | |||
 | +1 (269) 372-8736 | |||
 | +1 (269) 372-3397 | |||
 | sales@anisyn.com | |||
| Chemical manufacturer | ||||
| Reddy Chemtech Inc. | USA | |||
|---|---|---|---|---|
 | www.reddychemtech.com | |||
 | +1 (404) 483-6270 | |||
 | +1 (770) 420-0884 | |||
 | sales@reddychemtech.com | |||
| Chemical manufacturer | ||||
| Molekula Ltd | UK | |||
|---|---|---|---|---|
 | www.molekula.com | |||
 | +44 (1747) 831-066 | |||
 | +44 (1747) 831-199 | |||
 | info@molekula.com | |||
| Chemical manufacturer | ||||
| Wako Pure Chemical Industries, Ltd. | Japan | |||
|---|---|---|---|---|
 | www.wako-chem.co.jp | |||
 | +81 (6) 6203-3741 | |||
 | +81 (6) 6201-5964 | |||
 | wkhk.info@fujifilm.com | |||
| Chemical manufacturer since 1922 | ||||
| Toronto Research Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.trc-canada.com | |||
 | +1 (416) 665-9696 | |||
 | +1 (416) 665-4439 | |||
 | info@trc-canada.com | |||
| Chemical manufacturer since 1982 | ||||
| Matrix Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.matrixscientific.com | |||
 | +1 (803) 788-9494 | |||
 | +1 (803) 788-9419 | |||
 | sales@matrixscientific.com | |||
| Chemical manufacturer | ||||
| Frontier Scientific Services, Inc. | USA | |||
|---|---|---|---|---|
 | www.frontierssi.com | |||
 | +1 (302) 266-6891 (888) 577-2734 | |||
 | +1 (302) 266-8296 | |||
 | customerservice@frontierssi.com | |||
| Chemical manufacturer | ||||
| SynChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.synchem.com | |||
 | +1 (847) 298-2436 ex 230 | |||
 | +1 (847) 298-2439 | |||
 | sales@synchem.com | |||
| Chemical manufacturer since 1997 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Bromopyrimidine |
|---|---|
| Name | 2-Amino-5-bromopyrimidine |
| Synonyms | 5-bromopyrimidin-2-amine |
| Molecular Structure | 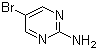 |
| Molecular Formula | C4H4BrN3 |
| Molecular Weight | 174.00 |
| CAS Registry Number | 7752-82-1 |
| EC Number | 629-581-4 |
| SMILES | C1=C(C=NC(=N1)N)Br |
| Density | 1.8±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 241-243 °C (Expl.) |
| Index of Refraction | 1.649, Calc.* |
| Boiling Point | 340.7±34.0 °C (760 mmHg), Calc.* |
| Flash Point | 159.9±25.7 °C, Calc.* |
| Water solubility | Insoluble (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS09 Warning Details GHS07;GHS09 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335-H400 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P273-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| Transport Information | UN 3077 | ||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
2-Amino-5-bromopyrimidine is a halogenated pyrimidine derivative, featuring a bromine atom at the 5-position and an amino group at the 2-position on the pyrimidine ring. Pyrimidine itself is a six-membered heterocyclic compound with nitrogen atoms at positions 1, 3, and 4, making it a core structure in many biologically active compounds, particularly nucleic acid analogs. The discovery of 2-amino-5-bromopyrimidine is significant due to its versatility in chemical synthesis and its role as an intermediate for various pharmacological and agricultural applications. The synthesis of 2-amino-5-bromopyrimidine typically involves the bromination of 2-amino-4,6-dihydroxypyrimidine or related precursors using brominating agents. This reaction selectively introduces the bromine atom at the 5-position, which is crucial for subsequent reactions that take advantage of the halogen’s reactivity. The presence of the amino group at the 2-position further enhances the reactivity of this compound, allowing it to undergo nucleophilic substitution reactions that are valuable in the synthesis of complex molecules. In organic synthesis, 2-amino-5-bromopyrimidine serves as an important intermediate for the construction of pyrimidine-based compounds. Its bromine substitution makes it an excellent candidate for cross-coupling reactions, such as Suzuki or Heck reactions, enabling the introduction of various functional groups. These reactions are particularly useful in the preparation of biologically active molecules, including pharmaceuticals and agrochemicals. The amino group at the 2-position also allows for the formation of derivatives through electrophilic substitution or condensation reactions, making 2-amino-5-bromopyrimidine a versatile building block in organic chemistry. One of the most prominent applications of 2-amino-5-bromopyrimidine is in the field of medicinal chemistry. Pyrimidine derivatives are a class of compounds with a wide range of biological activities, including antitumor, antiviral, and antimicrobial properties. The bromine atom in 2-amino-5-bromopyrimidine plays a key role in enhancing the compound's interaction with biological targets. For instance, the compound has been studied for its potential as an anticancer agent, with research indicating that bromine substitution can increase the binding affinity of pyrimidine derivatives to enzymes involved in cell division and DNA replication. Moreover, the amino group can be involved in hydrogen bonding interactions with biological macromolecules, potentially improving the pharmacokinetics of the compound. In agrochemicals, 2-amino-5-bromopyrimidine is used as a precursor for the synthesis of herbicides and fungicides. Pyrimidine derivatives have shown effectiveness in controlling plant diseases and pests due to their ability to inhibit specific enzymes in microorganisms or plants. The bromine substitution in 2-amino-5-bromopyrimidine enhances the molecule's stability and bioactivity, making it an attractive candidate for agricultural applications. Additionally, the amino group facilitates the formation of derivatives that can be optimized for selective activity against particular pests or diseases, contributing to more targeted and efficient crop protection. In material science, the halogenated pyrimidine derivatives, including 2-amino-5-bromopyrimidine, are being explored for their use in organic semiconductors. The unique electronic properties of pyrimidine rings, combined with the influence of the bromine atom, make these compounds suitable for applications in organic electronics, such as organic light-emitting diodes (OLEDs) and organic photovoltaics (OPVs). The halogen atom can modify the charge transfer properties of the compound, improving its performance in electronic devices. In conclusion, 2-amino-5-bromopyrimidine is a highly versatile compound with significant potential in various fields, including organic synthesis, medicinal chemistry, agrochemicals, and material science. The combination of a reactive bromine atom and an amino group at distinct positions on the pyrimidine ring makes this compound a valuable intermediate for the preparation of more complex molecules. As research progresses, the applications of 2-amino-5-bromopyrimidine in drug development, crop protection, and electronic materials are expected to expand, highlighting its importance in both industrial and scientific endeavors. References 1973. Mass spectra and structure of halo-substituted 2- and 4-aminopyrimidines. Chemistry of Heterocyclic Compounds. DOI: 10.1007/bf00477573 2009. Synthesis of Pyridine, Pyrimidine and Pyridinone C-Nucleoside Phosphoramidites for Probing Cytosine Function in RNA. The Journal of Organic Chemistry. DOI: 10.1021/jo9016919 2008. Acetylation of N-Heteroaryl Bromides via PdCl2/(o-tolyl)3P Catalyzed Heck Reactions. Synthesis. DOI: 10.1055/s-2008-1032193 |
| Market Analysis Reports |