Fosfomycin tromethamine is the tromethamine (often “trometamol”) salt of Fosfomycin, a broad-spectrum phosphonic antibiotic originally discovered in 1969 from a soil-derived micro-organism. Its molecular formula (free acid form) is C₃H₇O₄P and it derives from epoxide-phosphonic acid chemistry. The tromethamine salt form is used for oral administration (for example single-dose therapy) due to improved solubility and pharmacokinetics.
The discovery of fosfomycin stemmed from the screening of actinomycete strains for antimicrobial activity; researchers at the Spanish company CEPA in collaboration with Merck discovered the producing strain (Streptomyces fradiae) and isolated the antibiotic (initially named phosphonomycin). Subsequent structural elucidation revealed its novel epoxide and phosphonate moieties, which act by irreversible inhibition of the enzyme MurA (UDP-N-acetylglucosamine enolpyruvyl transferase), blocking the first committed step in bacterial peptidoglycan biosynthesis. This unique mechanism offers activity against both Gram-positive and Gram-negative bacteria and minimal cross-resistance with other antibiotic classes.
The application of fosfomycin tromethamine has been most prominent in the treatment of uncomplicated urinary tract infections (UTIs), especially in women. A single 3 g oral dose of the tromethamine salt yields high urinary concentrations of the active drug for 24 to 48 h, enabling convenient outpatient therapy. Clinical reviews report bacteriological eradication rates of 75-90 % in acute cystitis and very favourable tolerance, giving the drug a convenient administration profile. More recently, because of rising antimicrobial resistance, fosfomycin has been revisited for off-label and salvage use in multidrug-resistant Gram-negative infections, complicated UTIs, bone and joint infections, and other serious infections, often in combination regimens. Despite its early origin, it remains relevant in modern antibiotic stewardship algorithms.
Pharmacokinetically, the tromethamine salt form of fosfomycin is rapidly absorbed (bioavailability around 30-40 % in the fasted state), with a plasma half-life of around 5.7 h and predominant renal excretion of unchanged drug. Its high urinary concentration, low protein binding and novel mechanism together support the single-dose regimen in uncomplicated UTI. Resistance can develop via reduced uptake (GlpT/UhpT transporters), modifying enzymes or target mutation, but rates remain low in many settings—enhancing its attractiveness in settings with limited oral antibiotic options.
From a therapeutic perspective, fosfomycin tromethamine exemplifies the concept of “old drug, new utility”. Its use as a single-dose oral treatment for lower urinary tract infections is well established; further, its expanding role as a partner in multidrug regimens, particularly against extended-spectrum beta-lactamase–producing Enterobacteriaceae and Pseudomonas spp., reflects its renewed importance. The ease of administration (especially single-sachet oral dosing), favourable safety profile and broad spectrum continue to support its inclusion in guidelines and antimicrobial policy.
In summary, fosfomycin tromethamine is a clinically and historically significant antibiotic. Its origin from soil-derived actinomycetes, unique enzymatic target (MurA inhibition), oral single-dose application in urinary infection and resurgence in resistance-driven therapy all highlight its sustained relevance. As antibiotic resistance pressures grow, it remains a valuable agent both in its established indication and in evolving roles.
References
Patel SS, Balfour JA & Bryson HM (1997) Fosfomycin tromethamine. A review of its antibacterial activity, pharmacokinetic properties and therapeutic efficacy as a single-dose oral treatment for acute uncomplicated lower urinary tract infections. Drugs 53(4) 637–56. DOI: 10.2165/00003495-199753040-00007
Michalopoulos AS & Falagas ME (2011) The revival of fosfomycin. International Journal of Antimicrobial Agents 38(2) 91–99. DOI: 10.1016/j.ijid.2011.07.007
Dijkmans AC, Zacarías N, Burggraaf J, Mouton JW, Van de Velde NM, Degener JE, Van der Meer JW, Buijs J (2017) Fosfomycin: pharmacological, clinical and future perspectives. International Journal of Antimicrobial Agents 50(1) 10–19. DOI: 10.3390/antibiotics6040024
|
































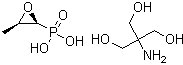

 GHS07;GHS08 Danger Details
GHS07;GHS08 Danger Details