Online Database of Chemicals from Around the World
| Changzhou Chenqiang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cq-chem.com | |||
 | +86 (519) 8890-2455 (510) 6689-6080 +86 18015336699 | |||
 | +86 (519) 8890-4599 | |||
 | info@cq-chem.com sales@wxichem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1996 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Minko Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.minkochem.com | |||
 | +86 (571) 5602-1871 | |||
 | +86 (571) 8720-9919 | |||
 | info@minkochem.com frank_vetpharm@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| The Dow Chemical Company | USA | |||
|---|---|---|---|---|
 | www.dow.com | |||
 | +1 (989) 633-1706 | |||
 | fmdcigs@dow.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| iFar Chemical Limited | China | |||
|---|---|---|---|---|
 | www.ifarchem.com | |||
 | +86 (510) 6689-6080 +86 18015336699 | |||
 | +86 (510) 8370-6633 | |||
 | sales@ifarchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 1998 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Kemai Chemical (Europe) Gmbh | Germany | |||
|---|---|---|---|---|
 | kemaichemgmbh.com | |||
 | +49 (495) 6456-3560 | |||
 | info@kemaichemgmbh.com | |||
| Chemical distributor since 1998 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Ozon Chemical | Poland | |||
|---|---|---|---|---|
 | ozonchemical.com | |||
 | +48 658908709 | |||
 | Info@ozonchemical.com | |||
| Chemical distributor since 1999 | ||||
| chemBlink Standard supplier since 2025 | ||||
| SelectLab Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.selectlab.de | |||
 | +49 (2383) 919-350 919-351 | |||
 | +49 (2383) 919-354 | |||
 | info@selectlab.de | |||
| Chemical manufacturer | ||||
| Jintan Kangyuan Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jtkychem.com | |||
 | +86 (519) 8256-6282 8256-6295 8269-9398 | |||
 | +86 (519) 8256-6281 / 8288-6169 | |||
 | sales@jtkychem.com | |||
| Chemical manufacturer since 1991 | ||||
| Classification | Chemical reagent >> Organic reagent >> Nitro compound |
|---|---|
| Name | Nitroethane |
| Synonyms | 1-nitroethane |
| Molecular Structure | 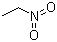 |
| Molecular Formula | C2H5NO2 |
| Molecular Weight | 75.07 |
| CAS Registry Number | 79-24-3 |
| EC Number | 201-188-9 |
| SMILES | CC[N+](=O)[O-] |
| Density | 1.0±0.1 g/cm3 Calc.*, 1.045 g/mL (Expl.) |
|---|---|
| Melting point | -90 °C (Expl.) |
| Boiling point | 109.2±3.0 °C 760 mmHg (Calc.)*, 114 - 115 °C (Expl.) |
| Flash point | 30.6 °C (Calc.)*, 31 °C (Expl.) |
| Solubility | 4.6 g/100 ml (20 °C) (Expl.) |
| Index of refraction | 1.383 (Calc.)*, 1.391 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS02;GHS07 Warning Details GHS02;GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H302-H332 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P261-P264-P270-P271-P280-P301+P317-P303+P361+P353-P304+P340-P317-P330-P370+P378-P403+P235-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2842 | ||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||
|
Nitroethane is a colorless liquid organic compound with the chemical formula C2H5NO2. It belongs to the class of nitroalkanes and is characterized by the presence of a nitro functional group (-NO2) attached to an ethane backbone. Nitroethane has a slightly sweet odor and is moderately soluble in water, with greater solubility in organic solvents such as alcohols and ethers. Nitroethane was first synthesized in the mid-19th century through the reaction of ethane derivatives with nitric acid or by the nitration of ethyl compounds. It is commercially produced by the vapor-phase nitration of propane or ethane, which involves reacting the hydrocarbons with nitric acid under controlled temperature and pressure conditions to yield nitroalkanes including nitroethane. This compound has significant industrial and laboratory applications. Nitroethane is widely used as a solvent for various chemical reactions and processes due to its ability to dissolve a wide range of compounds. It serves as a precursor and building block in organic synthesis, particularly in the manufacture of pharmaceuticals, agrochemicals, and dyes. Nitroethane is often employed in the synthesis of amphetamines and other compounds that require a nitroalkane intermediate. In the field of fuel technology, nitroethane is used as a component in high-performance racing fuels. Its ability to provide extra oxygen during combustion increases power output, making it valuable in drag racing and model engine applications. Nitroethane’s combustion properties allow engines to operate with higher efficiency and increased power compared to conventional hydrocarbons. Due to its chemical reactivity, nitroethane can participate in various organic transformations such as nucleophilic substitutions, reductions, and condensations. It acts as a nitroalkane nucleophile in Henry (nitroaldol) reactions, enabling the formation of carbon-carbon bonds, which are important in synthetic organic chemistry. Safety and environmental considerations are important in the handling of nitroethane. It is classified as a flammable and potentially toxic substance. Exposure to nitroethane vapors can cause irritation of the respiratory tract, headaches, and dizziness. Prolonged or high-level exposure may lead to more severe health effects. Appropriate protective measures, including adequate ventilation and personal protective equipment, are necessary when working with nitroethane. Environmental impact from nitroethane is generally related to its volatility and potential for air pollution. Proper storage, handling, and disposal procedures are required to minimize risks to both human health and the environment. Nitroethane continues to be a valuable chemical in industrial applications and research, combining solvent properties, synthetic utility, and fuel performance enhancement in various fields. References 2025. Retro-aza-Henry-type conversion of β-nitrostyrenes to the related hydrazones using ascorbic acid-functionalized graphene as a catalyst. Journal of the Iranian Chemical Society, 22(3). DOI: 10.1007/s13738-025-03194-z 2024. Effects of the structure of primary nitroalkanes and reaction conditions on the selectivity of acylamination of arenes in polyphosphoric acid. Russian Chemical Bulletin, 73(5). DOI: 10.1007/s11172-024-4277-8 2023. NiZnFe2O4: an eco-compatible and magnetically separable catalyst for multicomponent synthesis of 2-amino-4H-chromenes. Research on Chemical Intermediates, 49(6). DOI: 10.1007/s11164-023-05043-1 |
| Market Analysis Reports |