Online Database of Chemicals from Around the World
| Haimen Best Fine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bestfine.com | |||
 | +86 (513) 8538-3385 +86 13506281858 | |||
 | +86 (513) 8538-3386 | |||
 | best@bestfine.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Labseeker Inc | USA | |||
|---|---|---|---|---|
 | www.labseeker.com | |||
 | +1 (858) 750-1632 | |||
 | +1 (858) 412-1220 | |||
 | marketing@labseeker.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Weifang Fine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qianjinchemical.com | |||
 | +86 (536) 517-0190 | |||
 | +86 (536) 889-7109 | |||
 | 1297429207@qq.com | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Anhui Royal Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.royal-chem.com | |||
 | +86 (25) 8663-9253 | |||
 | +86 (25) 5227-9193 | |||
 | zoey.zhao@royal-chem.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Lavin Chemical (nantong) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lavinchem.com | |||
 | +86 13862887077 | |||
 | lavin@lavinchem.com | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Hebei Anlin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.anlinpharma.com | |||
 | +86 (310) 806-7016 806-5018 | |||
 | +86 (310) 806-5018 | |||
 | info@anlinpharma.com | |||
| Chemical manufacturer since 2002 | ||||
| Qidong A&P Chemical Factory Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.a-pchem.cn | |||
 | +86 (513) 8388-3000 8388-4000 8388-5000 | |||
 | +86 (513) 8388-6000 / 8388-7000 | |||
 | exp@a-pchem.com | |||
| Chemical manufacturer | ||||
| ChemPur GmbH | Germany | |||
|---|---|---|---|---|
 | www.chempur.de | |||
 | +49 (721) 933-840 | |||
 | +49 (721) 47-2001 | |||
 | info@chempur.de | |||
| Chemical manufacturer | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| Whyte Chemicals | UK | |||
|---|---|---|---|---|
 | www.whytechemicals.co.uk | |||
 | +44 (20) 8346-5946 | |||
 | +44 (20) 8349-4589 | |||
 | sales@whytechemicals.co.uk | |||
| Chemical distributor | ||||
| Zhao Du Fine Chemical Plant | China | |||
|---|---|---|---|---|
 | www.zdchem.com | |||
 | +86 (310) 404-1664 | |||
 | +86 (310) 404-9808 | |||
 | info@zdchem.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Chemical pesticide >> Insecticide intermediate |
|---|---|
| Name | Oxalyl chloride |
| Synonyms | Oxalyl dichloride; Ethanedioyl dichloride |
| Molecular Structure | 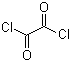 |
| Molecular Formula | C2Cl2O2 |
| Molecular Weight | 126.93 |
| CAS Registry Number | 79-37-8 |
| EC Number | 201-200-2 |
| SMILES | C(=O)(C(=O)Cl)Cl |
| Density | 1.6±0.1 g/cm3 Calc.*, 1.5 g/mL (Expl.) |
|---|---|
| Melting point | -10 - -8 °C (Expl.) |
| Boiling point | 63.5±9.0 °C 760 mmHg (Calc.)*, 62 - 65 °C (Expl.) |
| Flash point | 4.5±19.3 °C (Calc.)* |
| Solubility | reacts (Expl.) |
| Index of refraction | 1.449 (Calc.)*, 1.429 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |     GHS02;GHS05;GHS06;GHS07 Danger Details GHS02;GHS05;GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H260-H301-H314-H318-H331-H335 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P223-P231+P232-P260-P261-P264-P264+P265-P270-P271-P280-P301+P316-P301+P330+P331-P302+P335+P334-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P319-P321-P330-P363-P370+P378-P402+P404-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2922 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Oxalyl chloride is an acyl chloride with the molecular formula C2Cl2O2. Structurally, it consists of a central carbon–carbon bond flanked by two carbonyl groups, each bonded to a chlorine atom, giving the formula (COCl)2. It is a colorless to pale yellow liquid with a pungent odor and is highly reactive, particularly toward nucleophiles and moisture. The compound reacts vigorously with water, producing carbon dioxide, carbon monoxide, and hydrochloric acid, and must therefore be handled under anhydrous conditions. Oxalyl chloride is primarily used as a reagent in organic synthesis. Its most common application is the conversion of carboxylic acids into acyl chlorides, which are highly reactive intermediates for forming esters, amides, and other carbonyl-containing compounds. This transformation is widely employed in both laboratory-scale and industrial chemical synthesis. The compound is also used in the synthesis of oxalate esters and other functionalized carbonyl compounds. Its ability to introduce reactive acyl chloride groups allows for sequential reactions in multistep organic syntheses. In combination with catalysts such as dimethylformamide (DMF), oxalyl chloride can efficiently convert carboxylic acids under mild conditions. Oxalyl chloride is typically prepared by the chlorination of oxalic acid or oxalates using phosphorus pentachloride or thionyl chloride. Due to its high reactivity and corrosive nature, handling requires strict adherence to safety protocols, including use of gloves, eye protection, and well-ventilated fume hoods. It is also highly flammable and should be stored away from moisture and heat. Overall, oxalyl chloride is a highly reactive acyl chloride widely used in organic synthesis for the preparation of acyl chlorides, esters, amides, and other carbonyl derivatives. Its structural simplicity, high electrophilicity, and ability to activate carboxylic acids make it an indispensable reagent in both academic and industrial chemistry. References 2010. Novel tricyclic pyrazole BRAF inhibitors with imidazole or furan central scaffolds. Bioorganic & Medicinal Chemistry, 18(18). DOI: 10.1016/j.bmc.2010.06.031 2005. Polymer-supported and polymeric chiral guanidines: Preparation and application to the asymmetric Michael reaction of iminoacetate with methyl vinyl ketone. Molecular Diversity, 9(4). DOI: 10.1007/s11030-005-8116-z |
| Market Analysis Reports |