Online Database of Chemicals from Around the World
| Chizhou Dongsheng Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dspharmchem.com | |||
 | +86 (576) 8429-2571 | |||
 | +86 (576) 8429-2560 | |||
 | ksz@dspharmchem.com | |||
| Chemical manufacturer since 1995 | ||||
| chemBlink Premium supplier since 2011 | ||||
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Isoquinoline compound |
|---|---|
| Name | 6-Isopropylisoquinoline |
| Synonyms | 6-(1-Methylethyl)isoquinoline |
| Molecular Structure | 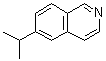 |
| Molecular Formula | C12H13N |
| Molecular Weight | 171.24 |
| CAS Registry Number | 790304-84-6 |
| SMILES | CC(C)C1=CC2=C(C=C1)C=NC=C2 |
| Solubility | Slightly soluble (1.1 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.026±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Melting point | 44-45 °C** |
| Boiling point | 284.1±9.0 °C (760 Torr), Calc.* |
| Flash point | 118.2±11.4 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2018 ACD/Labs) |
| ** | Gao, Mingzhang |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
6-Isopropylisoquinoline is a notable chemical compound with diverse applications in medicinal chemistry and natural product synthesis. This compound belongs to the isoquinoline family, which consists of a nitrogen-containing heterocyclic structure derived from benzene and pyridine. The introduction of an isopropyl group at the 6-position of the isoquinoline ring enhances its chemical properties and broadens its applications. The discovery of 6-Isopropylisoquinoline is attributed to the ongoing exploration of isoquinoline derivatives in organic chemistry. Isoquinolines are known for their presence in various natural products and their biological activities, which include antitumor, antibacterial, and anti-inflammatory properties. The modification of the isoquinoline ring through alkylation, such as the addition of an isopropyl group, has been a subject of interest due to the potential changes in pharmacological activity and the synthesis of novel compounds. 6-Isopropylisoquinoline is synthesized through several methods, typically involving the alkylation of isoquinoline with isopropylating agents. One common approach involves the use of Friedel-Crafts alkylation reactions, where isoquinoline reacts with isopropyl halides in the presence of a Lewis acid catalyst. This method allows for the selective introduction of the isopropyl group at the 6-position of the isoquinoline ring. In terms of applications, 6-Isopropylisoquinoline plays a significant role in the development of new pharmaceuticals and bioactive compounds. The presence of the isopropyl group can influence the compound's interaction with biological targets, potentially leading to enhanced activity or selectivity. For instance, isoquinoline derivatives have been studied for their potential as enzyme inhibitors, receptor modulators, and anticancer agents. The introduction of the isopropyl group can affect the molecule's lipophilicity and binding affinity, making it a valuable modification in drug design. Furthermore, 6-Isopropylisoquinoline serves as a useful intermediate in the synthesis of complex natural products and pharmaceutical agents. Its chemical structure allows for further functionalization, leading to the creation of a variety of isoquinoline-based compounds with tailored properties. This makes it an important building block in medicinal chemistry and organic synthesis. Research into the biological activities of 6-Isopropylisoquinoline and its derivatives continues to be an area of interest. Studies have shown that isoquinoline derivatives can exhibit a range of biological activities, including neuroprotective, anti-inflammatory, and antimicrobial effects. The specific impact of the isopropyl group on these activities is an ongoing topic of investigation, with the goal of developing new therapeutic agents with improved efficacy and safety profiles. In summary, 6-Isopropylisoquinoline is a valuable compound in the field of organic chemistry and pharmacology. Its synthesis and applications in drug development and natural product chemistry highlight its significance in creating new bioactive molecules and advancing medicinal research. References none |
| Market Analysis Reports |