Online Database of Chemicals from Around the World
| BrightGene Bio-medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bright-gene.com | |||
 | +86 (512) 6255-1801 6255-1767 +86 13812696362 | |||
 | +86 (512) 6255-1799 | |||
 | kevinwan@bright-gene.com kevinwan0203@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Min-biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.min-biotech.com | |||
 | +86 15190045345 | |||
 | sales@min-biotech.com | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Zhejiang Hisun Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hisunpharm.com | |||
 | +86 13651899693 | |||
 | yiwen.sun@hisunpharm.com | |||
| Chemical manufacturer since 1956 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | API >> Antineoplastic agents |
|---|---|
| Name | N2'-Deacetyl-N2'-(4-mercapto-4-methyl-1-oxopentyl)-maytansine |
| Synonyms | Maytansine DM 4; DM 4; Ravtansine;[(1S,2R,3S,5S,6S,16E,18E,20R,21S)-11-chloro-21-hydroxy-12,20-dimethoxy-2,5,9,16-tetramethyl-8,23-dioxo-4,24-dioxa-9,22-diazatetracyclo[19.3.1.110,14.03,5]hexacosa-10,12,14(26),16,18-pentaen-6-yl] (2S)-2-[methyl-(4-methyl-4-sulfanylpentanoyl)amino]propanoate |
| Molecular Structure | 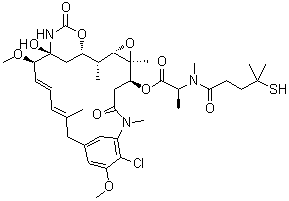 |
| Molecular Formula | C38H54ClN3O10S |
| Molecular Weight | 780.37 |
| CAS Registry Number | 796073-69-3 |
| EC Number | 641-067-1 |
| SMILES | C[C@@H]1[C@@H]2C[C@]([C@@H](/C=C/C=C(/CC3=CC(=C(C(=C3)OC)Cl)N(C(=O)C[C@@H]([C@]4([C@H]1O4)C)OC(=O)[C@H](C)N(C)C(=O)CCC(C)(C)S)C)C)OC)(NC(=O)O2)O |
| Solubility | Insoluble (3.7E-4 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.29±0.1 g/cm3 (20 °C 760 Torr), Calc.* |
| Melting point | 185-187 °C (decomp)** |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2015 ACD/Labs) |
| ** | Widdison, Wayne C. |
| Hazard Symbols |     GHS05;GHS06;GHS07;GHS08 Danger Details GHS05;GHS06;GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H300-H301-H310-H311-H314-H315-H319-H331-H340-H360-H361-H371 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P260-P261-P262-P264-P264+P265-P270-P271-P280-P301+P316-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P351+P338-P305+P354+P338-P308+P316-P316-P318-P321-P330-P332+P317-P337+P317-P361+P364-P362+P364-P363-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
N2'-Deacetyl-N2'-(4-mercapto-4-methyl-1-oxopentyl)-maytansine, commonly referred to as DM4, emerged from research efforts in the late 1970s by scientists at Takeda Chemical Industries in Japan. It is a potent cytotoxic agent derived from maytansine, a compound initially isolated from the plant Maytenus serrata. Through chemical modifications of maytansine's structure, DM4 was synthesized with the aim of enhancing its anticancer properties while reducing systemic toxicity. DM4 serves as the cytotoxic payload in ADCs, a class of targeted cancer therapies. In ADCs, DM4 is conjugated to monoclonal antibodies specific to antigens expressed on the surface of cancer cells. Upon binding to the target antigen, the ADC is internalized into the cancer cell, where DM4 is released and exerts its cytotoxic effects. This targeted delivery minimizes systemic toxicity and enhances the therapeutic index of DM4, making it an attractive option for cancer treatment.DM4-based ADCs have demonstrated efficacy in the treatment of solid tumors, including breast cancer, gastric cancer, and bladder cancer. ADCs such as trastuzumab deruxtecan (DS-8201) utilize DM4 as the cytotoxic payload and have shown promising results in clinical trials. DM4-based ADCs have also shown promise in the treatment of hematological malignancies. Several ADCs targeting different antigens expressed on hematological cancer cells, such as CD30 and CD22, incorporate DM4 as the cytotoxic payload. These ADCs have demonstrated efficacy in the treatment of Hodgkin lymphoma, non-Hodgkin lymphoma, and acute lymphoblastic leukemia, among others. References 2024. Synthesis and evaluation of antibody-drug conjugates with high drug-to-antibody ratio using dimaleimide-DM1 as a linker-payload. Bioorganic Chemistry, 149(1). DOI: 10.1016/j.bioorg.2024.107504 2024. eSOMA-DM1, a Maytansinoid-Based Theranostic Small-Molecule Drug Conjugate for Neuroendocrine Tumors. Bioconjugate Chemistry, 35(10). DOI: 10.1021/acs.bioconjchem.4c00413 1977. Tumor inhibitors. 122. The maytansinoids. Isolation, structural elucidation, and chemical interrelation of novel ansa macrolides. The Journal of Organic Chemistry, 42(14). DOI: 10.1021/jo00434a001 |
| Market Analysis Reports |