Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Yantai Topway Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.topway.biz | |||
 | +86 (535) 661-5990 | |||
 | +86 (535) 662-5679 | |||
 | yttopway@163.com | |||
| Chemical manufacturer since 1998 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Changchun BC&HC Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bchc-china.com | |||
 | +86 (431) 8087-1788 8087-1588 +86 15804318207 | |||
 | +86 (431) 8841-7879 | |||
 | 3468242165@qq.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Sinocure Chemical Group | China | |||
|---|---|---|---|---|
 | www.sinocurechem.com | |||
 | +86 15550440621 | |||
 | info@sinocurechem.com | |||
| Chemical manufacturer since 2020 | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Organic sulfur compound |
|---|---|
| Name | Cumyl hydroperoxide |
| Synonyms | 1-Mmethyl-1-phenylethylhydroperoxide; alpha,alpha-Dimethylbenzylhydroperoxide; Cumene hydroperoxide |
| Molecular Structure | 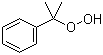 |
| Molecular Formula | C9H12O2 |
| Molecular Weight | 152.19 |
| CAS Registry Number | 80-15-9 |
| EC Number | 201-254-7 |
| SMILES | CC(C)(C1=CC=CC=C1)OO |
| Density | 1.038 |
|---|---|
| Melting point | -30 °C |
| Boiling point | 153 °C |
| Flash point | 83 °C |
| Water solubility | Slightly soluble |
| Hazard Symbols |      GHS02;GHS06;GHS08;GHS05;GHS09 Danger Details GHS02;GHS06;GHS08;GHS05;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H242-H302-H312-H314-H317-H318-H331-H341-H373-H411 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P210-P234-P235-P240-P260-P261-P264-P264+P265-P270-P271-P272-P273-P280-P301+P317-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P318-P319-P321-P330-P333+P317-P362+P364-P363-P370+P378-P391-P403-P403+P233-P405-P410-P411-P420-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2116; UN 3109 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Cumyl hydroperoxide, also known as 1-phenyl-1-(peroxy)propan-2-ol, is a chemical compound with significant industrial and research applications. It was first synthesized in the early 20th century and has since become an important reagent in various chemical processes. The discovery of cumyl hydroperoxide can be traced back to the early work on peroxides and hydroperoxides, which are known for their reactivity and role as oxidizing agents. Cumyl hydroperoxide was developed as part of research into new peroxide compounds that could be used in polymerization and oxidation reactions. Its unique properties, including its stability and ability to decompose to release free radicals, made it a valuable tool in chemical synthesis. Cumyl hydroperoxide is primarily used as a polymerization initiator. In polymer chemistry, it serves as a radical initiator in the polymerization of various monomers, including styrene and acrylates. Its ability to generate free radicals upon decomposition makes it an effective catalyst for initiating polymerization reactions. This application is crucial in the production of plastics, resins, and other polymeric materials, where controlled polymerization is necessary to achieve desired material properties. In addition to its role in polymerization, cumyl hydroperoxide is used in the oxidation of organic compounds. It acts as an oxidizing agent in various chemical reactions, including the oxidation of alkenes to ketones and aldehydes. This application is valuable in synthetic organic chemistry, where precise oxidation reactions are required to produce specific chemical compounds. Cumyl hydroperoxide also finds use in the synthesis of other chemicals. It is employed in the production of certain pharmaceuticals and agrochemicals, where its reactivity and ability to introduce functional groups into molecules are utilized. Its role in these processes highlights its versatility and importance in industrial chemistry. Despite its utility, cumyl hydroperoxide must be handled with care due to its reactive nature. It is a strong oxidizing agent and can be hazardous if not managed properly. Safety precautions are necessary to prevent accidents and ensure safe handling in industrial and laboratory settings. Overall, cumyl hydroperoxide is a valuable chemical with diverse applications in polymer chemistry, oxidation reactions, and chemical synthesis. Its discovery and development have made significant contributions to various fields, demonstrating its importance in both industrial and research contexts. References 1994. Lipid hydroperoxides evoke antigonototropic and antisteroidogenic activity in rat luteal cells. Endocrinology, 135(6). DOI: 10.1210/en.135.6.2723 1984. Hydroperoxides as inactivators of aromatase: 10.beta.-hydroperoxy-4-estrene-3,17-dione, crystal structure and inactivation characteristics. Biochemistry, 23(22). DOI: 10.1021/bi00318a004 2024. Triphenylphosphine in the identification of oxidation products of organic compounds with molecular oxygen and peroxides. Russian Chemical Bulletin, 73(9). DOI: 10.1007/s11172-024-4367-7 |
| Market Analysis Reports |