Online Database of Chemicals from Around the World
| Chirogate International Inc. | Taiwan | |||
|---|---|---|---|---|
 | www.chirogate.com | |||
 | +886 (3) 496-3808 | |||
 | +886 (3) 496-3800 | |||
 | annyeh@chirogate.com kirogate@ms63.hinet.net | |||
| Chemical manufacturer since 2000 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Foreland Pharma Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.forelandpharma.com | |||
 | +86 (10) 5631-5231 5631-5237 | |||
 | +86 (10) 5631-5231 | |||
 | qi.ji@forelandpharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Nanjing Xize Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.njxizebio.com | |||
 | +86 (25) 6602-3220 | |||
 | +86 (25) 6602-3220 | |||
 | sale@njxizebio.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Genriver Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.genriverpharm.com | |||
 | +86 13761582449 +86 13482015261 | |||
 | +86 (21) 3778-2903 | |||
 | info@genriverpharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Turtle Pharma Private Limited | India | |||
|---|---|---|---|---|
 | www.turtlepharma.com | |||
 | +91 9850998504 | |||
 | info@turtlepharma.com | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Cayman Chemical Company | USA | |||
|---|---|---|---|---|
 | www.caymanchem.com | |||
 | +1 (734) 971-3335 | |||
 | +1 (734) 971-3640 | |||
 | sales@caymanchem.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Hormone and endocrine-regulating drugs >> Prostaglandins |
|---|---|
| Name | Remodulin |
| Synonyms | Treprostinil; [[(1R,2R,3aS,9aS)-2,3,3a,4,9,9a-Hexahydro-2-hydroxy-1-[(3S)-3-hydroxyoctyl]-1H-benz[f]inden-5-yl]oxy]acetic acid |
| Molecular Structure | 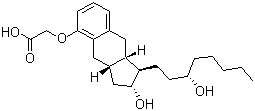 |
| Molecular Formula | C23H34O5 |
| Molecular Weight | 390.51 |
| CAS Registry Number | 81846-19-7 |
| EC Number | 808-233-7 |
| SMILES | CCCCC[C@@H](CC[C@H]1[C@@H](C[C@H]2[C@@H]1CC3=C(C2)C(=CC=C3)OCC(=O)O)O)O |
| Solubility | 100 mM (DMSO), 50 mM (ethanol) |
|---|---|
| Hazard Symbols |    GHS06;GHS07;GHS08 Danger Details GHS06;GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H311-H315-H319-H331-H335-H361-H372 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P260-P261-P262-P264-P264+P265-P270-P271-P280-P301+P316-P302+P352-P304+P340-P305+P351+P338-P316-P318-P319-P321-P330-P332+P317-P337+P317-P361+P364-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
Remodulin, also known by its generic name treprostinil, is a pharmaceutical drug used to treat pulmonary arterial hypertension (PAH), a serious condition characterized by high blood pressure in the arteries of the lungs. PAH can lead to heart failure if left untreated, and Remodulin plays a crucial role in managing this condition by helping to lower pulmonary blood pressure and improve exercise capacity. Treprostinil, the active ingredient in Remodulin, is a synthetic analogue of prostacyclin, a naturally occurring substance in the body that helps to dilate blood vessels and prevent blood clotting. Prostacyclin's role in vascular homeostasis made it a target for drug development aimed at treating PAH. Remodulin was developed as a more stable and longer-acting alternative to earlier prostacyclin-based therapies, which required continuous intravenous administration due to their short half-lives. Remodulin was approved for medical use in the early 2000s, following extensive research and clinical trials that demonstrated its efficacy and safety in patients with PAH. Unlike its predecessors, Remodulin can be administered via subcutaneous or intravenous infusion, offering greater flexibility and convenience for patients. The subcutaneous route, in particular, allows for continuous drug delivery using a small pump, which can significantly improve patients' quality of life by reducing the need for frequent hospital visits. The mechanism of action of Remodulin involves vasodilation, which helps to reduce the resistance in the pulmonary arteries, thereby lowering blood pressure in the lungs. Additionally, Remodulin inhibits platelet aggregation, reducing the risk of blood clots that can further complicate PAH. These combined effects make Remodulin a critical component of PAH treatment, particularly for patients who are unable to tolerate other therapies or whose condition has progressed despite standard treatments. Remodulin is typically prescribed as part of a long-term management plan for PAH and is often used in conjunction with other medications, such as endothelin receptor antagonists or phosphodiesterase-5 inhibitors, to optimize treatment outcomes. The dosing of Remodulin is individualized based on the patient's response and tolerability, with the goal of gradually increasing the dose to achieve maximum therapeutic benefit while minimizing side effects. Common side effects of Remodulin include infusion site pain and reactions, headache, nausea, diarrhea, and jaw pain. These side effects are generally manageable and may diminish over time as the patient adjusts to the therapy. However, the administration method, especially the subcutaneous route, can sometimes cause significant discomfort at the infusion site, which may limit its use in some patients. Despite these challenges, Remodulin has been a transformative treatment for many individuals with PAH, helping to extend survival, improve symptoms, and enhance quality of life. The development of treprostinil-based therapies continues, with newer formulations and delivery methods being explored to further improve patient outcomes. References 2021. Inhaled Treprostinil in Pulmonary Hypertension Due to Interstitial Lung Disease. The New England journal of medicine, 384(4). DOI: 10.1056/nejmoa2008470 2003. Prostanoids for Pulmonary Arterial Hypertension. American journal of respiratory medicine : drugs, devices, and other interventions, 2(2). DOI: 10.1007/bf03256644 2012. Inhaled treprostinil: a therapeutic review. Drug Design, Development and Therapy, 6. DOI: 10.2147/dddt.s19281 |
| Market Analysis Reports |