Online Database of Chemicals from Around the World
| CAS: 82208-84-2 Product: 3-Allylfentanyl No suppliers available. |
| Classification | Organic raw materials >> Heterocyclic compound >> Piperidines |
|---|---|
| Name | 3-Allylfentanyl |
| Synonyms | N-phenyl-N-[1-(2-phenylethyl)-3-prop-2-enylpiperidin-4-yl]propanamide |
| Molecular Structure | 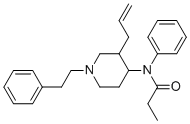 |
| Molecular Formula | C25H32N2O |
| Molecular Weight | 376.53 |
| CAS Registry Number | 82208-84-2 |
| SMILES | CCC(=O)N(C1CCN(CC1CC=C)CCC2=CC=CC=C2)C3=CC=CC=C3 |
| Density | 1.1±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 494.9±38.0 °C 760 mmHg (Calc.)* |
| Flash point | 189.8±19.1 °C (Calc.)* |
| Index of refraction | 1.568 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
|
3-Allylfentanyl is a synthetic opioid analgesic belonging to the 4-anilidopiperidine class and is structurally related to fentanyl. It features a substitution at the 3-position of the piperidine ring with an allyl group, distinguishing it from the parent compound. Like other fentanyl analogs, 3-allylfentanyl exhibits high affinity for the μ-opioid receptor, leading to potent analgesic effects along with a risk of severe respiratory depression. The compound was originally developed in the context of research exploring structure-activity relationships among fentanyl derivatives. Modifications at various positions on the core fentanyl scaffold have been studied to evaluate their impact on receptor binding affinity, pharmacokinetics, and side effect profiles. The introduction of an allyl group at the 3-position in 3-allylfentanyl alters both the lipophilicity and steric interactions at the receptor site. This change can influence potency, efficacy, and duration of action. Although 3-allylfentanyl has demonstrated opioid activity in preclinical settings, it has not been developed for clinical use. One possible reason is the narrow therapeutic index common to fentanyl analogs, which increases the risk of unintentional overdose. Another factor is that small changes in structure can lead to unpredictable pharmacodynamic and pharmacokinetic profiles, making such compounds unsuitable for routine therapeutic application without extensive testing. In forensic toxicology, 3-allylfentanyl has been identified as a designer opioid appearing in illicit drug markets. The rise of novel psychoactive substances has led to the emergence of a wide variety of fentanyl analogs, often synthesized and distributed to bypass legal controls. In response, regulatory agencies have taken measures to control 3-allylfentanyl and related compounds under analogue or temporary scheduling laws. Analytical detection of 3-allylfentanyl in biological samples requires sensitive instrumentation due to the compound’s high potency and typically low concentrations in forensic contexts. Methods such as gas chromatography-mass spectrometry (GC-MS) and liquid chromatography-tandem mass spectrometry (LC-MS/MS) are commonly employed to confirm its presence and differentiate it from structurally similar analogs. Reference materials are essential for accurate identification and quantification, particularly in clinical toxicology and postmortem investigations. The biological effects of 3-allylfentanyl are consistent with those of other μ-opioid receptor agonists, including profound analgesia, euphoria, sedation, and respiratory depression. At higher doses or in opioid-naive individuals, it can rapidly induce life-threatening respiratory compromise. Naloxone remains an effective opioid antagonist for reversing its effects, but higher or repeated dosing may be required due to the compound’s potency. From a regulatory standpoint, compounds like 3-allylfentanyl are typically controlled under legislation targeting fentanyl analogs. Many countries have implemented broad structural class-based scheduling frameworks to preemptively regulate such compounds before they gain widespread illicit use. These legal measures are designed to reduce the public health risks associated with the rapid proliferation of synthetic opioids. 3-Allylfentanyl serves as an example of how slight structural modifications to known opioid scaffolds can produce substances with potent pharmacological activity and significant safety concerns. It underscores the importance of continued monitoring, research, and regulation of emerging synthetic opioids to mitigate their potential impact on public health and safety. References 1982. 3-Allyl analogues of fentanyl. The Journal of Pharmacy and Pharmacology, 34(3). DOI: 10.1111/j.2042-7158.1982.tb04229.x PMID: 6121908 |
| Market Analysis Reports |