Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Sigma-Aldrich, Inc. | China | |||
|---|---|---|---|---|
 | www.sigmaaldrich.com | |||
 | +86 (21) 6141-5566 800-819-3336 | |||
 | orderCN@sial.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Changzhou Comwin Fine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.comwin-china.com | |||
 | +86 (519) 8661-9955 | |||
 | +86 (519) 8661-3190 | |||
 | info@comwin-china.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2024 | ||||
| SynInnova Laboratories Inc. | USA | |||
|---|---|---|---|---|
 | www.syninnova.com | |||
 | +1 (780) 485-2962 | |||
 | +1 (780) 485-2963 | |||
 | sales@syninnova.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Imidazoles |
|---|---|
| Name | 4-Methyl-2-phenyl-1H-imidazole |
| Synonyms | 5-methyl-2-phenyl-1H-imidazole |
| Molecular Structure | 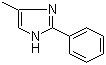 |
| Molecular Formula | C10H10N2 |
| Molecular Weight | 158.20 |
| CAS Registry Number | 827-43-0 |
| EC Number | 212-571-5 |
| SMILES | CC1=CN=C(N1)C2=CC=CC=C2 |
| Melting point | 180-183 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
4-Methyl-2-phenyl-1H-imidazole is an organic compound that belongs to the imidazole family, characterized by its unique five-membered ring structure with nitrogen atoms at positions 1 and 3. This substance has garnered attention for its versatile chemical properties, as well as its applications across several fields including materials science, pharmaceuticals, and organic synthesis. The discovery and exploration of imidazole derivatives like 4-Methyl-2-phenyl-1H-imidazole emerged from early 20th-century research aimed at understanding the reactivity and utility of imidazoles in various chemical reactions. The synthesis of 4-Methyl-2-phenyl-1H-imidazole involves the reaction of aldehydes or ketones with amines in the presence of an acid catalyst, a process that has been refined over time to allow for higher yields and more efficient production. The addition of a methyl group at the fourth position and a phenyl group at the second position of the imidazole ring significantly alters the compound’s chemical properties, enhancing its stability and providing it with new functional capabilities. This combination of a phenyl group and a methyl substituent makes it a valuable intermediate in various chemical transformations. One of the main applications of 4-Methyl-2-phenyl-1H-imidazole is in the field of pharmaceuticals, where it serves as a building block for the synthesis of bioactive molecules. Imidazole-based structures are well-known for their pharmacological activity, particularly as antimicrobial, antifungal, and anti-inflammatory agents. 4-Methyl-2-phenyl-1H-imidazole is often utilized in drug design as a core structure to develop compounds that target specific biological receptors. The presence of the phenyl group increases lipophilicity, which can enhance the ability of drug molecules to penetrate biological membranes and achieve better therapeutic efficacy. In addition to its pharmaceutical applications, 4-Methyl-2-phenyl-1H-imidazole is also employed in materials science. Imidazole derivatives, in general, have proven useful as curing agents for epoxy resins, which are widely used in coatings, adhesives, and composite materials. The imidazole structure acts as a catalyst in the polymerization process, allowing for faster curing and improving the mechanical properties of the resulting epoxy. The methyl and phenyl groups in 4-Methyl-2-phenyl-1H-imidazole can enhance its performance in these roles, making it a valuable additive in high-performance applications such as aerospace and electronics. Moreover, the compound is a key intermediate in the synthesis of complex organic molecules. It is often used in chemical reactions that require imidazole-based ligands for catalysis. These ligands are involved in a wide range of organic reactions, including transition metal-catalyzed processes such as cross-coupling reactions. The structure of 4-Methyl-2-phenyl-1H-imidazole provides the necessary electron-donating and steric properties to facilitate these reactions, making it a preferred choice in laboratory synthesis and industrial-scale production. Another notable use of 4-Methyl-2-phenyl-1H-imidazole is in the development of corrosion inhibitors. Imidazole derivatives are known for their ability to form protective layers on metal surfaces, thus preventing oxidation and corrosion. In industrial settings where machinery and equipment are exposed to harsh environmental conditions, additives such as 4-Methyl-2-phenyl-1H-imidazole are used to extend the service life of these materials. From a safety perspective, 4-Methyl-2-phenyl-1H-imidazole should be handled with care, as it can cause irritation to the skin, eyes, and respiratory system upon exposure. Proper protective measures, including gloves, goggles, and adequate ventilation, are recommended when working with this compound. Despite these safety precautions, the compound’s broad utility makes it an indispensable tool for chemists and engineers alike. In summary, 4-Methyl-2-phenyl-1H-imidazole is a versatile chemical compound with a wide range of applications, particularly in the pharmaceutical, materials science, and industrial sectors. Its imidazole-based structure, combined with the functional groups attached at key positions, enhances its reactivity and makes it a valuable intermediate for many chemical processes. As research into imidazole derivatives continues, 4-Methyl-2-phenyl-1H-imidazole is likely to remain an important component in both commercial and academic chemistry. References 2023. The Novel Organic Emitters for High-Performance Narrow-Band Deep Blue OLEDs. Topics in current chemistry (Cham), 381(5). DOI: 10.1007/s41061-023-00436-7 2019. New trends in corrosion protection of copper. Chemical Papers, 73(9). DOI: 10.1007/s11696-019-00774-1 2012. Theoretical investigations on the molecular structure and vibrational spectral analysis of 4-methyl 2-phenylimidazole. Optics and Spectroscopy, 113(6). DOI: 10.1134/s0030400x12090032 |
| Market Analysis Reports |