Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Nanjing Finetech Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fine-chemtech.com | |||
 | +86 (25) 5207-8417 +86 17714198479 | |||
 | +86 (25) 5207-8417 | |||
 | sales@fine-chemtech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2007 | ||||
| ZHIYU Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zhiyubiotech.com | |||
 | +86 (512) 6279-1916 | |||
 | +86 (512) 6279-1915 | |||
 | sales@zhiyubiotech.com info@zhiyubiotech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Beijing Eagle Sky Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.eagleskypharmatech.com | |||
 | +86 (10) 5979-9429 8875-5821 | |||
 | +86 (10) 5804-3698 | |||
 | sophia_818@126.com contact@eagleskypharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Ich Biofarm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ichemie.com | |||
 | +86 (571) 2818-6870 | |||
 | +86 (571) 8993-9479 | |||
 | specialchem1@ichemie.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Rochi Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rochipharma.com | |||
 | +86 (21) 3875-1876 +86 15000076078 | |||
 | +86 (21) 5027-5764 | |||
 | info@rochipharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2015 | ||||
| NANJING TOMMLAB PHARMATECH Co., LTD | China | |||
|---|---|---|---|---|
 | www.tommlab.com | |||
 | +86 (025) 8616-1360 | |||
 | tommlab@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Pure Bio Medical Jiangsu Ltd. | China | |||
|---|---|---|---|---|
 | www.purejs.cn | |||
 | +86 (025) 8560-7383 | |||
 | jiangziwei@purejs.cn | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | API >> Other chemicals |
|---|---|
| Name | Elagolix sodium |
| Synonyms | NBI 56418NA |
| Molecular Structure | 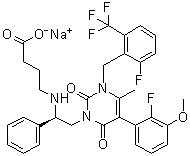 |
| Molecular Formula | C32H29F5N3O5.Na |
| Molecular Weight | 653.57 |
| CAS Registry Number | 832720-36-2 |
| EC Number | 692-765-8 |
| SMILES | CC1=C(C(=O)N(C(=O)N1CC2=C(C=CC=C2F)C(F)(F)F)C[C@@H](C3=CC=CC=C3)NCCCC(=O)[O-])C4=C(C(=CC=C4)OC)F.[Na+] |
| Hazard Symbols |   GHS07;GHS08 Danger Details GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H304-H319-H360-H361 Details | ||||||||||||||||||||||||||||
| Safety Statements | P203-P264+P265-P280-P301+P316-P305+P351+P338-P318-P331-P337+P317-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Elagolix sodium is the sodium salt form of elagolix, an orally active, non-peptide gonadotropin-releasing hormone (GnRH) receptor antagonist. It is used primarily in the management of hormone-dependent conditions such as endometriosis and uterine fibroids. The molecular formula of elagolix sodium is C32H29F3N4NaO5, reflecting its complex structure that includes fluorinated aromatic rings and heterocyclic components designed for receptor binding and bioavailability. Elagolix was developed to provide a more flexible and reversible suppression of the hypothalamic-pituitary-gonadal axis compared to traditional GnRH agonists. By competitively binding to GnRH receptors in the pituitary gland, elagolix sodium inhibits the release of luteinizing hormone (LH) and follicle-stimulating hormone (FSH), leading to a decrease in ovarian sex hormone production, mainly estrogen and progesterone. This hormonal suppression alleviates symptoms caused by estrogen-dependent diseases such as pelvic pain associated with endometriosis. The compound's discovery emerged from efforts to develop small-molecule GnRH antagonists with improved oral bioavailability and safety profiles. Elagolix sodium's design incorporates modifications to optimize receptor affinity, metabolic stability, and pharmacokinetic properties allowing once or twice daily oral dosing. Pharmacokinetically, elagolix sodium is rapidly absorbed after oral administration, with peak plasma concentrations typically achieved within one to two hours. It is metabolized primarily by cytochrome P450 enzymes, particularly CYP3A4, and is excreted mainly via the feces. The drug has a half-life of approximately four to six hours, supporting its dosing schedule. Clinically, elagolix sodium is indicated for the management of moderate to severe pain associated with endometriosis and, in some regions, for the treatment of heavy menstrual bleeding associated with uterine fibroids. Its ability to reduce estrogen levels without causing complete hypoestrogenism offers an advantage in minimizing side effects such as bone loss and vasomotor symptoms. Adverse effects reported with elagolix sodium use include hot flushes, headache, nausea, and decreased bone mineral density with long-term use. Monitoring is recommended during therapy, especially in patients with risk factors for osteoporosis. In summary, elagolix sodium is an orally active GnRH receptor antagonist designed to modulate reproductive hormone levels for the treatment of estrogen-dependent gynecological conditions. Its chemical structure and pharmacological profile provide effective symptom relief with a manageable safety profile. References 2008. Discovery of Sodium R-(+)-4-{2-[5-(2-Fluoro-3-methoxyphenyl)-3-(2-fluoro-6-[trifluoromethyl]benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl]-1-phenylethylamino}butyrate (Elagolix), a Potent and Orally Available Nonpeptide Antagonist of the Human Gonadotropin-Releasing Hormone Receptor. Journal of Medicinal Chemistry, 51(23). DOI: 10.1021/jm8006454 2009. Suppression of Gonadotropins and Estradiol in Premenopausal Women by Oral Administration of the Nonpeptide Gonadotropin-Releasing Hormone Antagonist Elagolix. The Journal of Clinical Endocrinology and Metabolism, 94(2). DOI: 10.1210/jc.2008-1695 2024. The short- and mid-term efficacy and safety of elagolix in the management of pain associated with endometriosis: A systematic review and meta-analysis. Journal of Gynecology Obstetrics and Human Reproduction, 53(7). DOI: 10.1016/j.jogoh.2024.102829 |
| Market Analysis Reports |