Online Database of Chemicals from Around the World
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Wonderchem Co., Limited | Hong Kong | |||
|---|---|---|---|---|
 | www.wonder-chem.com | |||
 | +852 8192-5218 | |||
 | sales@wonder-chem.com | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Classification | Chemical reagent >> Organic reagent >> Phosphine ligand |
|---|---|
| Name | Josiphos SL-J216-2 |
| Synonyms | (2S)-1-[(1S)-1-[Bis(1,1-dimethylethyl)phosphino]ethyl]-2-(di-1-naphthalenylphosphino)ferrocene |
| Molecular Structure | 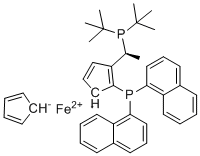 |
| Molecular Formula | C40H44FeP2 |
| Molecular Weight | 642.57 |
| CAS Registry Number | 849924-44-3 |
| SMILES | [Fe+2].C[C@@H](C1C=C[CH-]C=1P(C1C=CC=C2C=CC=CC2=1)C1C=CC=C2C=CC=CC2=1)P(C(C)(C)C)C(C)(C)C.[CH-]1C=CC=C1 |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
Josiphos SL-J216-2, a prominent member of the Josiphos family of ligands, has made substantial contributions to the field of organometallic chemistry since its introduction. This compound is a chiral phosphine ligand notable for its ability to influence the reactivity and selectivity of transition metal-catalyzed reactions. The synthesis of Josiphos SL-J216-2 involves a carefully controlled reaction of phosphine derivatives with chiral bidentate ligands. The preparation process requires precise conditions to ensure the formation of the desired ligand with high purity. The ligand’s structure is characterized by a phosphorus atom bonded to two distinct chiral groups, creating a chiral environment that significantly affects its interaction with metal centers. Josiphos SL-J216-2 was developed to address the need for efficient chiral ligands in asymmetric catalysis. Its design incorporates features that provide enhanced steric and electronic properties, which are essential for controlling the stereochemistry of reactions. This makes it particularly valuable for applications where high enantioselectivity is required. One of the primary applications of Josiphos SL-J216-2 is in asymmetric hydrogenation. The ligand's ability to stabilize transition metal centers, such as those in rhodium or ruthenium complexes, enables it to facilitate the hydrogenation of various substrates with high enantioselectivity. This application is crucial for the synthesis of chiral pharmaceuticals and fine chemicals where precise control over stereochemistry is required. In addition to hydrogenation, Josiphos SL-J216-2 has been used in other asymmetric transformations, including hydroformylation and allylic substitution reactions. Its effectiveness in these reactions is attributed to its ability to influence the electronic and steric environment of the metal center, thereby enhancing the selectivity and activity of the catalytic process. The versatility of Josiphos SL-J216-2 extends beyond traditional organic synthesis. It has been explored for its potential applications in material science and catalysis, where its chiral properties can be leveraged to develop new materials with specific optical or electronic properties. Overall, Josiphos SL-J216-2 exemplifies the advancements in ligand design for asymmetric catalysis. Its development has provided chemists with a valuable tool for achieving high enantioselectivity in a range of reactions, furthering the capabilities of organometallic chemistry and its applications in both industrial and academic settings. References none |
| Market Analysis Reports |