Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wuhan Xinlaibokang Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xblkchem.com | |||
 | +86 18202741975 | |||
 | 3762793534@qq.com | |||
 | QQ Chat | |||
 | WeChat: 18202741975 | |||
| Chemical manufacturer since 2021 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | API >> Other chemicals |
|---|---|
| Name | Mirodenafil |
| Synonyms | 5-Ethyl-3,5-dihydro-2-[5-[[4-(2-hydroxyethyl)-1-piperazinyl]sulfonyl]-2-propoxyphenyl]-7-propyl-4H-pyrrolo[3,2-d]pyrimidin-4-one |
| Molecular Structure | 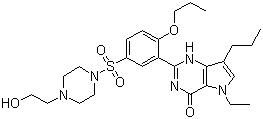 |
| Molecular Formula | C26H37N5O5S |
| Molecular Weight | 531.67 |
| CAS Registry Number | 862189-95-5 |
| SMILES | CCCC1=CN(C2=C1N=C(NC2=O)C3=C(C=CC(=C3)S(=O)(=O)N4CCN(CC4)CCO)OCCC)CC |
| Solubility | 10 mM (DMSO) |
|---|---|
| Density | 1.33 |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319 Details |
| Safety Statements | P501-P270-P264-P280-P302+P352-P337+P313-P305+P351+P338-P362+P364-P332+P313-P301+P312+P330 Details |
|
Mirodenafil is a potent pharmaceutical compound primarily known for its application in treating erectile dysfunction (ED). Discovered and developed by the South Korean company SK Chemicals, Mirodenafil represents a significant advancement in the field of urology and sexual health. The discovery of Mirodenafil came as part of a broader effort to develop selective and effective phosphodiesterase type 5 (PDE5) inhibitors. PDE5 inhibitors are a class of drugs that work by blocking the enzyme phosphodiesterase type 5, which plays a role in regulating blood flow in the penis. By inhibiting this enzyme, Mirodenafil helps increase blood flow to the penis, facilitating and maintaining an erection in response to sexual stimulation. Mirodenafil was developed with the intention of providing a therapeutic option with improved efficacy and a favorable side effect profile compared to existing PDE5 inhibitors such as sildenafil (Viagra), tadalafil (Cialis), and vardenafil (Levitra). The chemical structure of Mirodenafil includes a pyrazolopyrimidinone core, which contributes to its high specificity and potency in inhibiting PDE5. Clinically, Mirodenafil has been shown to be effective in treating ED in various populations, including those with underlying health conditions like diabetes and hypertension. It is typically administered in tablet form, with dosages tailored to individual patient needs. Studies have demonstrated that Mirodenafil has a rapid onset of action, usually within 30 to 60 minutes, and a duration of effect that allows for flexibility in sexual activity. One of the notable advantages of Mirodenafil is its safety and tolerability profile. Clinical trials and post-marketing surveillance have shown that it is generally well-tolerated, with common side effects being mild and transient, including headaches, flushing, nasal congestion, and dyspepsia. These side effects are similar to those associated with other PDE5 inhibitors but tend to be less severe and less frequent. Beyond its primary use in treating ED, Mirodenafil has also been explored for other potential medical applications. Research is ongoing into its possible benefits in treating pulmonary arterial hypertension (PAH), a condition characterized by high blood pressure in the arteries of the lungs. The mechanism of action of PDE5 inhibitors, which promotes vasodilation and improved blood flow, makes them promising candidates for PAH treatment. Additionally, the development of Mirodenafil underscores the importance of pharmaceutical innovation in improving patient outcomes and quality of life. Its introduction into the market has provided another valuable option for individuals suffering from ED, contributing to a broader range of therapeutic choices and personalized treatment approaches. References 2007. Development of LC/MS/MS assay for the determination of 5-ethyl-2-{5-[4-(2-hydroxyethyl)piperazine-1-sulfonyl]-2-propoxyphenyl}-7-propyl-3,5-dihydropyrrolo[3,2-d]pyrimidin-4-one (SK3530) in human plasma: application to a clinical pharmacokinetic study. Journal of Pharmaceutical and Biomedical Analysis, 45(2). DOI: 10.1016/j.jpba.2007.06.021 2007. Validation of a HPLC method for the quantification and purity determination of SK3530 in drug substance and tablet. Journal of Pharmaceutical and Biomedical Analysis, 43(3). DOI: 10.1016/j.jpba.2006.10.011 2007. Metabolism and excretion of 5-ethyl-2-{5-[4-(2-hydroxyethyl)piperazine-1-sulfonyl]-2-propoxyphenyl}-7-propyl-3,5-dihydropyrrolo[3,2-d]-pyrimidin-4-one (SK3530) in rats. Rapid Communications in Mass Spectrometry, 21(6). DOI: 10.1002/rcm.2943 |
| Market Analysis Reports |