Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Nanjing Finetech Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fine-chemtech.com | |||
 | +86 (25) 5207-8417 +86 17714198479 | |||
 | +86 (25) 5207-8417 | |||
 | sales@fine-chemtech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Biocompounds Pharmaceutical Inc. | China | |||
|---|---|---|---|---|
 | www.biocompounds.com | |||
 | +86 (21) 5768-7675 | |||
 | +86 (21) 5768-7169 | |||
 | sales@biocompounds.com info@biocompounds.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Shanghai Boc Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bocchem.com | |||
 | +86 (21) 3497-5602 +86 15618693615 | |||
 | +86 (21) 3497-5603 | |||
 | sales@bocchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Hangzhou StarShine Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.starshinepharm.com | |||
 | +86 (571) 8512-3681 +86 13777804878 | |||
 | +86 (571) 8512-2157 | |||
 | sales@starshinepharm.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Bengbu Nako Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.nakochem.com | |||
 | +86 (552) 306-8842 | |||
 | sales@nakochem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Nanjing Bic Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bicbiotech.com | |||
 | +86 18851183712 | |||
 | sales@bicbiotech.com | |||
 | QQ Chat | |||
 | WeChat: +8618851183712 | |||
 | WhatsApp:+8618851183712 | |||
| Chemical manufacturer since 2023 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | Organic raw materials >> Amino compound >> Amide compound |
|---|---|
| Name | (E)-N-(2-Chloro-6-methylphenyl)-3-ethoxyacrylamide |
| Synonyms | (2E)-N-(2-Chloro-6-methylphenyl)-3-ethoxy-2-propenamide |
| Molecular Structure | 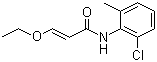 |
| Molecular Formula | C12H14ClNO2 |
| Molecular Weight | 239.70 |
| CAS Registry Number | 863127-76-8 |
| SMILES | CCO/C=C/C(=O)NC1=C(C=CC=C1Cl)C |
| Density | 1.2$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 386.5$+/-$42.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 187.6$+/-$27.9 $degree$C (Calc.)* |
| Index of refraction | 1.573 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
(E)-N-(2-Chloro-6-methylphenyl)-3-ethoxyacrylamide is a substituted aromatic acrylamide in which a benzene ring is functionalized with a chloro group at the 2-position, a methyl group at the 6-position, and an acrylamide moiety bearing an ethoxy substituent at the β-position. Compounds of this type are relevant in medicinal chemistry and organic synthesis due to their α,β-unsaturated carbonyl system, which can act as a Michael acceptor, and the combination of aromatic and polar functionalities that modulate electronic properties and reactivity. The (E)-configuration of the double bond provides defined geometry, which can influence biological activity and chemical selectivity. Structurally, the molecule consists of a substituted aniline derivative linked via an amide bond to an ethoxy-substituted α,β-unsaturated carbonyl group. The chlorine atom is electron-withdrawing, affecting the electronic density of the aromatic ring, while the methyl group provides steric modulation and lipophilicity. The acrylamide portion features a conjugated C=C–C=O system, with the ethoxy substituent on the β-carbon increasing solubility and slightly modifying electrophilicity. The (E)-configuration ensures that the substituents on the double bond are in trans orientation, influencing both steric and electronic properties in reactions and potential biological interactions. The synthesis of (E)-N-(2-chloro-6-methylphenyl)-3-ethoxyacrylamide is typically achieved through condensation of 2-chloro-6-methylaniline with an ethyl-substituted acyl chloride or ester derivative under basic or catalytic conditions to form the acrylamide linkage. Control of temperature and solvent is critical to favor the (E)-isomer and to prevent polymerization of the α,β-unsaturated system. Purification yields a stable crystalline compound suitable for further chemical use. In medicinal chemistry, α,β-unsaturated acrylamides are often explored as enzyme inhibitors or covalent modifiers due to the Michael acceptor nature of the double bond. The aromatic substituents can engage in π–π stacking or hydrophobic interactions, while the amide linkage allows hydrogen bonding with target residues. The combination of the electron-withdrawing chlorine, lipophilic methyl group, and the ethoxy-modified acrylamide provides a scaffold suitable for modulating reactivity and binding affinity. Such compounds can be investigated for activity against kinases, proteases, or other nucleophilic biological targets. Beyond pharmaceutical applications, this compound serves as an intermediate in organic synthesis for generating more complex α,β-unsaturated amides or heterocyclic derivatives. The conjugated system can undergo Michael additions, cycloadditions, or further functionalization at the β-carbon, while the aromatic substituents allow selective modifications through halogen or methyl chemistry. This makes it useful for creating libraries of derivatives for structure–activity studies or material applications. Overall, (E)-N-(2-chloro-6-methylphenyl)-3-ethoxyacrylamide is a multifunctional substituted acrylamide combining an α,β-unsaturated carbonyl system with aromatic chlorine and methyl substituents and a β-ethoxy group. Its combination of Michael acceptor reactivity, hydrogen-bonding potential, and aromatic electronic modulation makes it a valuable intermediate for organic synthesis, medicinal chemistry, and the development of bioactive compounds. References 2017. Substituted pyridines as inhibitors of DNMT1. KR-20190017030-A. DOI: 10.1007/s10593-019-02570-x 2018. Preparation method of dasatinib. CN-109265455-B. DOI: https://patents.google.com/patent/CN109265455B/en 2015. Crystalline forms of n-(2-chloro-6-methylphenyl)-2-[[6-[4-(2-hydroxyethyl)-1-piperazinyl]-2-methyl-4-pyrimidinyl]amino]-5-thiazolecarboxamide and their process thereof. WO-2017002131-A1. DOI: https://patents.google.com/patent/WO2017002131A1/en |
| Market Analysis Reports |