Online Database of Chemicals from Around the World
| Suzhou Biosyntech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.biosyntech-suzhou.com | |||
 | +86 13921151340 +86 (512) 6300-1269 | |||
 | sales2@biosyntech-suzhou.com sales@biosyntech-suzhou.com | |||
 | QQ Chat | |||
| Chemical distributor since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Imidazoles |
|---|---|
| Name | methyl 2-methyl-2-(4-nitro-1H-imidazol-1-yl)propanoate |
| Molecular Structure | 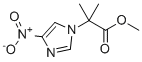 |
| Molecular Formula | C8H11N3O4 |
| Molecular Weight | 213.19 |
| CAS Registry Number | 865774-07-8 |
| SMILES | CC(C)(C(=O)OC)N1C=C(N=C1)[N+](=O)[O-] |
| Density | 1.3±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 377.8±22.0 °C 760 mmHg (Calc.)* |
| Flash point | 182.3±22.3 °C (Calc.)* |
| Index of refraction | 1.559 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P280-P301+P312-P302+P352-P305+P351+P338 Details |
| SDS | Available |
|
Methyl 2-methyl-2-(4-nitro-1H-imidazol-1-yl)propanoate is a small organic compound that features a substituted imidazole ring connected to a tertiary alkyl ester. This molecule is chemically related to compounds with known biological activity, particularly within the class of nitroimidazole derivatives, many of which are widely studied for their antimicrobial and radiosensitizing properties. The core structural feature of this molecule is the 4-nitro-1H-imidazole ring. Imidazole is a five-membered heterocyclic ring containing two nitrogen atoms at non-adjacent positions. The presence of a nitro group at the 4-position of the ring enhances the compound's electron-withdrawing properties, which is often crucial for redox activity in biological environments. Nitroimidazoles have been extensively investigated due to their ability to undergo bioreductive activation under hypoxic conditions. This property makes them especially valuable in targeting hypoxic regions in tumors or anaerobic microorganisms, where their reduction leads to cytotoxic intermediates capable of DNA damage. The nitroimidazole ring is N-substituted with a tertiary alkyl ester moiety: a 2-methyl-2-propanoate ester in which the central carbon is bonded to two methyl groups and a carboxylic ester function. The full name of this side chain is methyl 2-methylpropanoate. The presence of two methyl groups on the α-carbon introduces steric bulk, enhancing metabolic stability and possibly affecting the molecule’s pharmacokinetic profile by hindering enzymatic hydrolysis. The ester group, on the other hand, increases the molecule's lipophilicity and can serve as a prodrug strategy, allowing passive diffusion across cellular membranes and enzymatic cleavage in vivo to release the active acid or alcohol. The molecular design reflects a balance between metabolic stability and bioactivation. While the nitroimidazole core can be selectively reduced in low-oxygen environments, the ester moiety may modulate the compound’s solubility, membrane permeability, and rate of clearance. In pharmaceutical applications, such a compound might be investigated for its potential in treating infections caused by anaerobic bacteria or protozoa, or for its use as a radiosensitizing agent in cancer therapy. Radiosensitizers are compounds that make tumor cells more susceptible to the effects of radiation, and nitroimidazoles have a history of clinical use in this domain. The synthesis of methyl 2-methyl-2-(4-nitro-1H-imidazol-1-yl)propanoate likely involves nucleophilic substitution at the N1 position of 4-nitroimidazole with a suitable alkylating agent such as methyl 2-bromo-2-methylpropanoate. This alkylation reaction can be conducted under basic conditions, where the deprotonated imidazole acts as a nucleophile and attacks the electrophilic carbon of the alkyl halide. The resulting N-substituted product would then contain the ester moiety directly linked to the imidazole ring. Compounds of this type may also serve as starting points for further derivatization in medicinal chemistry programs. Modifications at the ester function, such as conversion to amides or acids, or at the nitro group, such as reduction to amines, could yield analogues with different activity profiles or improved selectivity. The nitro group itself is a pharmacophore of interest, but it is also associated with potential toxicity concerns, so the balance between efficacy and safety must be carefully evaluated. In drug design, the inclusion of a nitroimidazole ring suggests a deliberate effort to exploit hypoxia-selective cytotoxicity or antimicrobial effects. The methylated ester side chain may have been introduced to enhance pharmacokinetic characteristics without significantly compromising the molecule’s biological reactivity. Given the known activities of similar nitroimidazole-containing molecules, such as metronidazole and misonidazole, compounds like methyl 2-methyl-2-(4-nitro-1H-imidazol-1-yl)propanoate may represent structurally simplified analogues or intermediates for the development of next-generation therapeutic agents targeting hypoxic tumors or anaerobic infections. References none |
| Market Analysis Reports |