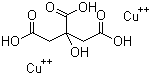
Copper citrate is a coordination compound formed from copper ions and citric acid. It exists in various hydrated and anhydrous forms and is recognized for its solubility properties and potential applications in fields such as catalysis, agriculture, and medicine. The coordination chemistry of copper with citrate ions has been studied since the early 20th century, highlighting the ability of citric acid to act as a multidentate ligand, binding metal ions through its carboxylate and hydroxyl groups.
The synthesis of copper citrate typically involves reacting copper salts such as copper sulfate or copper nitrate with citric acid in aqueous solution, often under controlled pH and temperature to favor complex formation. The resulting copper citrate complex is generally isolated as a crystalline solid, which can vary in hydration state depending on the preparation conditions. Structural analyses have revealed that copper ions in copper citrate complexes adopt coordination geometries influenced by the citrate ligand’s flexibility, often leading to chelation and polymeric network structures.
Copper citrate has been investigated for its antimicrobial properties, attributed to the combined effects of copper ions and the organic ligand. Its controlled solubility enhances bioavailability, making it a subject of interest in agricultural applications as a micronutrient fertilizer to correct copper deficiencies in plants. In catalysis, copper citrate complexes have been utilized as catalysts or catalyst precursors in oxidation reactions, benefiting from copper’s redox activity and the ligand’s stabilizing influence.
In medical research, copper citrate has been studied as a dietary supplement to address copper deficiency disorders. The bioavailability and reduced toxicity compared to other copper salts make copper citrate an attractive candidate for supplementation. Furthermore, studies have explored its potential antioxidant effects, owing to the metal-ligand interactions that modulate copper’s reactivity.
Handling and preparation of copper citrate require consideration of factors such as pH, ligand-to-metal ratio, and temperature to ensure reproducible synthesis and stability. The compound’s multifunctional properties continue to motivate research into its applications in sustainable agriculture, catalysis, and health.
References
2011. Goldfrank's Toxicologic Emergencies, 9th Ed. New York: McGraw-Hill, p. 1259.
|



























 GHS07;GHS09 Warning Details
GHS07;GHS09 Warning Details