Online Database of Chemicals from Around the World
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Sigma-Aldrich, Inc. | China | |||
|---|---|---|---|---|
 | www.sigmaaldrich.com | |||
 | +86 (21) 6141-5566 800-819-3336 | |||
 | orderCN@sial.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Van Aroma | Indonesia | |||
|---|---|---|---|---|
 | www.vanaroma.com | |||
 | +62 (21) 867-7003 | |||
 | +62 (21) 867-7002 | |||
 | marketing@vanaroma.com | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Jiangxi Zhonghuan New Materials Co., Ltd | China | |||
|---|---|---|---|---|
 | www.jxzhxcl.com | |||
 | 13766287383 | |||
 | 2246992030@qq.com | |||
 | QQ Chat | |||
 | WeChat: 13766287383 | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Jiangxi Xinsen Natural Vegetable Oil Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jxxinsen.com | |||
 | +86 18679667531 | |||
 | 592432494@qq.com | |||
 | QQ Chat | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Jiangxi Cedar Natural Medicinal Oil Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jxxs8-1.com | |||
 | +86 18000738807 | |||
 | 2134411736@qq.com | |||
 | QQ Chat | |||
 | WeChat: 592432494 | |||
| Chemical distributor since 2010 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Ji'an Guoguang Fragrance Factory | China | |||
|---|---|---|---|---|
 | www.jxggxlc.com | |||
 | +86 18607064978 | |||
 | 2529415945@qq.com | |||
 | QQ Chat | |||
| Chemical distributor since 1988 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Flavors and spices >> Synthetic spice >> Hydrocarbon spice >> Olefin |
|---|---|
| Name | L-Caryophyllene |
| Synonyms | trans-Caryophyllene; trans-beta-Caryophyllene; beta-Caryophyllen; beta-Caryophyllene; (-)-trans-Caryophyllene; (-)-beta-Caryophyllene; (1R,4E,9S)-4,11,11-Trimethyl-8-methylenebicyclo[7.2.0]undec-4-ene |
| Molecular Structure | 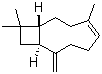 |
| Molecular Formula | C15H24 |
| Molecular Weight | 204.35 |
| CAS Registry Number | 87-44-5 |
| EC Number | 201-746-1 |
| SMILES | C/C/1=CCCC(=C)[C@H]2CC([C@@H]2CC1)(C)C |
| Density | 0.89 |
|---|---|
| Boiling point | 268 °C |
| Refractive index | 1.499 |
| Flash point | 105 °C |
| Hazard Symbols |   GHS07;GHS08 Danger Details GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H304-H317 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P272-P280-P301+P316-P302+P352-P321-P331-P333+P317-P362+P364-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
The discovery of L-caryophyllene dates back to the early 1800s, when scientists began to isolate and study essential oils. Beta-caryophyllene was first isolated from clove oil by German chemist Friedrich Wilhelm Sertürner in the early 19th century. Later, its chemical structure was elucidated as C15H24, which features a bicyclic structure with a cyclobutane ring—uncommon in natural compounds. L-caryophyllene is a colorless to pale yellow liquid with a woody, spicy odor. Its structure includes double bonds and a unique cyclobutane ring that contributes to its stability and reactivity. This sesquiterpene is able to interact with cannabinoid receptors in the human body, which further distinguishes it from other terpenes and is particularly attractive for therapeutic uses. L-caryophyllene is widely used in the flavor and fragrance industry for its rich aroma and ability to enhance the flavor of various food and beverage products. Its spicy, peppery flavor adds depth to savory dishes, sauces, and marinades. In the fragrance industry, its warm, woody scent is an important ingredient in formulating perfumes, colognes, and aromatherapy products, adding to their sophistication and appeal. In addition to its sensory contributions, L-caryophyllene has antimicrobial properties that help preserve the freshness and quality of food products. This makes it a beneficial addition to natural food preservation strategies. The most promising application of L-caryophyllene is its potential therapeutic function. It has anti-inflammatory properties due to its interaction with the CB2 cannabinoid receptor. Unlike THC (tetrahydrocannabinol), L-caryophyllene does not produce psychoactive effects, making it a safe anti-inflammatory and analgesic option. Research suggests that L-caryophyllene may help manage pain by modulating inflammatory responses and interacting with pain receptors. This makes it a hot topic for the development of natural pain relief alternatives, especially for conditions such as arthritis and neuropathic pain. Preliminary research suggests that L-caryophyllene may have anxiolytic and antidepressant effects. By affecting the endocannabinoid system, it helps relieve symptoms of anxiety and depression and promotes mental health without the side effects associated with traditional treatments. Its role in protecting the gastrointestinal tract from inflammation and oxidative stress has also gained attention, opening avenues for its use in treating conditions such as inflammatory bowel disease (IBD) and other gastrointestinal disorders. In agriculture, L-caryophyllene is a natural insect repellent. Its strong aroma repels a variety of insects, reducing the need for synthetic pesticides and promoting sustainable agricultural practices. Plants produce L-caryophyllene as part of their defense mechanism against herbivores and pathogens. Understanding its role in plant biology could help develop crop varieties with enhanced resistance to pests and diseases. References 2025. Investigation of the in vitro effects of cannabidiol, cannabidiolic acid, and the terpene β-caryophyllene on lymphocytes harvested from atopic and healthy dogs: A preliminary study. Research in Veterinary Science. DOI: 10.1016/j.rvsc.2024.105483 2024. Select terpenes from Cannabis sativa are antinociceptive in mouse models of post-operative pain and fibromyalgia via adenosine A2a receptors. Pharmacological reports : PR. DOI: 10.1007/s43440-024-00687-1 2008. Beta-caryophyllene is a dietary cannabinoid. Proceedings of the National Academy of Sciences, 105(26). DOI: 10.1073/pnas.0803601105 |
| Market Analysis Reports |