Online Database of Chemicals from Around the World
| Zibo Feitian International Trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.feitianindustry.com | |||
 | +86 (533) 276-3996 | |||
 | info@feitianindustry.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Premium supplier since 2022 | ||||
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridine compound >> Pyridine derivative |
|---|---|
| Name | 1,2,4,5-Benzenetetracarboxylic anhydride |
| Synonyms | Pyromellitic dianhydride; Benzene-1,2,4,5-tetracarboxylic dianhydride; PMDA |
| Molecular Structure | 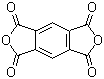 |
| Molecular Formula | C10H2O6 |
| Molecular Weight | 218.12 |
| CAS Registry Number | 89-32-7 |
| EC Number | 201-898-9 |
| SMILES | C1=C2C(=CC3=C1C(=O)OC3=O)C(=O)OC2=O |
| Density | 1.68 g/mL |
|---|---|
| Melting point | 283-287 °C |
| Boiling point | 397-400 °C |
| Flash point | 380 °C |
| Water solubility | decomposes |
| Hazard Symbols |    GHS05;GHS07;GHS08 Danger Details GHS05;GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H317-H318-H334 Details | ||||||||||||||||||||||||||||
| Safety Statements | P233-P260-P261-P264+P265-P271-P272-P280-P284-P302+P352-P304+P340-P305+P354+P338-P317-P321-P333+P317-P342+P316-P362+P364-P403-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
1,2,4,5-Benzenetetracarboxylic anhydride, commonly referred to as pyromellitic anhydride (PMA), is a significant compound in organic chemistry known for its versatile applications in various fields, including polymer science, coatings, and pharmaceuticals. Its unique structure, featuring four carboxylic acid groups, allows it to participate in a wide range of chemical reactions, making it an important building block for synthesizing other chemical entities. The discovery of pyromellitic anhydride dates back to the late 19th century when it was first synthesized by the oxidative degradation of certain aromatic compounds. Its anhydride form is particularly notable for its ability to react with various nucleophiles, resulting in the formation of useful derivatives. This reactivity has paved the way for its application in creating polymers and resins, particularly in the production of high-performance thermosetting materials. PMA serves as a key monomer in the synthesis of polyimides and other advanced materials, which exhibit excellent thermal stability and mechanical properties. In the realm of polymer chemistry, pyromellitic anhydride is often utilized as a hardener or crosslinking agent. When combined with epoxy resins, it enhances the thermal and chemical resistance of the final product. This property makes PMA an essential component in the formulation of coatings, adhesives, and composite materials, which are widely used in aerospace, automotive, and electronic applications. The incorporation of PMA into these materials significantly improves their durability, making them suitable for demanding environments. Another significant application of pyromellitic anhydride lies in the synthesis of various pharmaceutical compounds. Its reactivity enables it to act as a building block for creating complex organic molecules, which can possess biological activity. Research has shown that derivatives of PMA exhibit antimicrobial, anti-inflammatory, and anticancer properties, highlighting its potential in drug development. The ability to modify the anhydride functional groups further allows chemists to tailor these compounds for specific therapeutic uses, leading to the discovery of new drug candidates. Additionally, pyromellitic anhydride plays a crucial role in the production of specialty chemicals and functional materials. Its derivatives are employed in the manufacturing of surfactants, dyes, and plasticizers, demonstrating its versatility beyond traditional applications. The environmental impact of PMA is also a subject of research, as the focus on sustainable practices in chemical manufacturing has prompted investigations into greener synthesis routes and the development of biodegradable materials. Safety considerations are essential when handling pyromellitic anhydride, as it can cause skin and respiratory irritation. Proper safety protocols should be in place to mitigate exposure risks during its production and application. Ongoing research into the health effects and environmental impact of PMA underscores the importance of responsible usage in both industrial and laboratory settings. In conclusion, 1,2,4,5-benzenetetracarboxylic anhydride is a crucial compound in organic chemistry with diverse applications in polymer science, pharmaceuticals, and specialty chemicals. Its discovery as a versatile building block has led to significant advancements in material science and drug development. The continued exploration of pyromellitic anhydride and its derivatives may lead to further innovations, enhancing its importance in various scientific and industrial fields. References 2008. Pyromellitic Diimides: Minimal Cores for High Mobility n-Channel Transistor Semiconductors. Journal of the American Chemical Society, 130(42). DOI: 10.1021/ja805746h 2010. Preparation of zwitterionic hybrid polymer and its application for the removal of heavy metal ions from water. Journal of Hazardous Materials, 179(1-3). DOI: 10.1016/j.jhazmat.2010.02.041 2018. Controlled Release of DEET Loaded on Fibrous Mats from Electrospun PMDA/Cyclodextrin Polymer. Molecules, 23(7). DOI: 10.3390/molecules23071694 |
| Market Analysis Reports |