Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Earlier HK Company Limited | China | |||
|---|---|---|---|---|
 | www.earlierchem.com | |||
 | +86 (10) 6142-6769 +86 18616907430 | |||
 | +86 (10) 6142-6769 | |||
 | info@earlierchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Changzhou Sanling Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sanlingchem.com | |||
 | +86 (519) 8873-1370 +86 13921039671 | |||
 | +86 (519) 8893-2529 | |||
 | tanxinsong@163.com sales@sanlingchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2013 | ||||
| Sigma-Aldrich, Inc. | China | |||
|---|---|---|---|---|
 | www.sigmaaldrich.com | |||
 | +86 (21) 6141-5566 800-819-3336 | |||
 | orderCN@sial.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Piperidines |
|---|---|
| Name | Dichloro(2,2,6,6-tetramethylpiperidinato)magnesate(1-) lithium (1:1) |
| Synonyms | TMPMgCl·LiCl; TMPMgCl·LiCl |
| Molecular Structure | 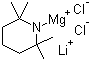 |
| Molecular Formula | C9H18Cl2MgN.Li |
| Molecular Weight | 242.40 |
| CAS Registry Number | 898838-07-8 |
| SMILES | [Li+].CC1(CCCC([N-]1)(C)C)C.[Mg+2].[Cl-].[Cl-] |
| Density | 0.96 g/mL (25 °C) (Expl.) |
|---|---|
| Flash point | -15 °C (Expl.) |
| Hazard Symbols |     GHS02;GHS08;GHS05;GHS07 Danger Details GHS02;GHS08;GHS05;GHS07 Danger Details |
|---|---|
| Risk Statements | H225-H314-H335-H336-H351-H361d Details |
| Safety Statements | P202-P210-P280-P303+P361+P353-P304+P340+P310-P305+P351+P338 Details |
| SDS | Available |
|
Dichloro(2,2,6,6-tetramethylpiperidinato)magnesate(1-) lithium (1:1) is a coordination complex combining lithium and magnesium. This compound represents a class of bimetallic organometallic reagents known for their unique reactivity and versatility in synthetic organic chemistry. Such species have become increasingly valuable in the development of reactions requiring controlled reactivity and specific selectivity, particularly in challenging bond-forming processes. The development of this compound builds on a foundation of research into mixed-metal reagents, which aim to exploit the complementary properties of two different metals. Magnesium, a common component in organometallic chemistry, is known for its nucleophilic character and wide applicability in Grignard-type reactions. Lithium, on the other hand, often enhances reactivity through its electropositive nature and ability to form diverse coordination structures. When combined, lithium and magnesium offer a synergistic framework, with lithium modifying the electronic and steric properties of the magnesium center. Dichloro(2,2,6,6-tetramethylpiperidinato)magnesate(1-) lithium is synthesized by reacting magnesium chloride with 2,2,6,6-tetramethylpiperidine in the presence of a lithium salt. The resulting complex exhibits unique properties due to the steric bulk and electron-donating characteristics of the tetramethylpiperidinate ligand. This bulky ligand stabilizes the reactive magnesium center while tuning its nucleophilicity and enabling selective transformations. The compound has found application in various organic transformations, particularly in the field of selective nucleophilic substitutions and deprotonation reactions. Its mild but effective reactivity makes it a valuable reagent in the preparation of sensitive intermediates that would otherwise decompose under harsher conditions. For example, it has been employed in the functionalization of heterocycles and the synthesis of organometallic intermediates that serve as precursors for complex natural products and pharmaceutical agents. One notable application of dichloro(2,2,6,6-tetramethylpiperidinato)magnesate(1-) lithium is in stereoselective synthesis, where control over the spatial arrangement of substituents is critical. The steric properties of the tetramethylpiperidinate ligand influence the approach of reactants, allowing for high selectivity in reactions such as alkylations and cross-couplings. This feature has proven advantageous in synthesizing biologically active molecules, where precise stereochemical configurations are crucial for activity. The compound also holds promise in the field of catalysis, where mixed-metal systems are increasingly explored for their ability to mediate complex transformations. Studies suggest that the unique coordination environment provided by this reagent could be utilized in cooperative catalysis, where both lithium and magnesium actively participate in reaction mechanisms. Dichloro(2,2,6,6-tetramethylpiperidinato)magnesate(1-) lithium exemplifies the potential of mixed-metal reagents in advancing synthetic methodologies. Its development and applications highlight the importance of ligand design and metal synergy in creating versatile and efficient chemical tools. References none |
| Market Analysis Reports |