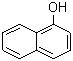
1-Naphthol, an aromatic organic compound with the formula C10H8O, is a derivative of naphthalene, characterized by a hydroxyl group attached to the first position of the naphthalene ring system. This compound is a white to yellow crystalline solid with a characteristic odor, and it plays a significant role in various chemical processes and applications across different industries. The discovery of 1-naphthol dates back to the early 19th century, with its first synthesis attributed to the German chemist Johann Wolfgang Döbereiner in 1820. Since then, it has been extensively studied and utilized for its versatile properties.
The primary industrial application of 1-naphthol lies in the production of dyes and pigments. Its ability to undergo electrophilic substitution reactions makes it an excellent precursor for various azo dyes. These dyes are widely used in textiles, food coloring, and cosmetics due to their vibrant colors and stability. The use of 1-naphthol in dye synthesis is particularly notable for producing acid dyes, which are used to color wool, silk, and nylon fibers. Additionally, it serves as a key ingredient in the production of fluorescent dyes, which have applications in biological imaging and diagnostics.
Apart from its role in dye production, 1-naphthol is also employed in the manufacturing of pesticides and herbicides. The compound's reactivity allows for the synthesis of various agrochemicals, which are vital for enhancing agricultural productivity. For instance, 1-naphthol derivatives are used in the formulation of insecticides and fungicides, contributing to pest management strategies in crop production. Its application in agrochemicals is essential for protecting crops from diseases and pests, ultimately improving food security.
Furthermore, 1-naphthol has found utility in the pharmaceutical industry. It acts as an important intermediate in the synthesis of various therapeutic compounds, including nonsteroidal anti-inflammatory drugs (NSAIDs) and analgesics. The compound's chemical structure allows for modifications that lead to the creation of biologically active molecules with potential medicinal properties. Research into the pharmacological effects of 1-naphthol and its derivatives has shown promise in developing new drugs for treating inflammatory conditions and other ailments.
In addition to its industrial and pharmaceutical applications, 1-naphthol is utilized in analytical chemistry. It serves as a reagent in various chemical reactions, including the synthesis of specific organic compounds and as a testing agent for the identification of certain metal ions. Its sensitivity to changes in pH and the presence of metal ions make it a valuable tool in environmental monitoring and chemical analysis.
Despite its numerous applications, safety considerations regarding the handling and use of 1-naphthol are essential. The compound can pose health risks if inhaled or ingested, and prolonged skin contact may lead to irritation. Therefore, proper safety measures, including personal protective equipment and ventilation, should be employed when working with 1-naphthol to mitigate exposure risks.
In summary, 1-naphthol is a significant organic compound with diverse applications in dye production, agrochemicals, pharmaceuticals, and analytical chemistry. Its discovery and subsequent utilization have contributed to advancements in various industries, highlighting its importance as a versatile chemical building block. Ongoing research into its properties and derivatives may lead to further innovations and applications, ensuring that 1-naphthol remains a vital compound in both industrial and scientific contexts.
References
1984. Metabolic activation of 1-naphthol by rat liver microsomes to 1,4-naphthoquinone and covalent binding species. Biochemical Pharmacology, 33(20).
DOI: 10.1016/0006-2952(84)90077-7
1970. Degradation of 1-naphthol in sea water. Journal of Agricultural and Food Chemistry, 18(1).
DOI: 10.1021/jf60167a035
1994. Radiation inactivation analysis of microsomal UDP-glucuronosyltransferases catalysing mono- and diglucuronide formation of 3,6-dihydroxybenzo(a)pyrene and 3,6-dihydroxychrysene. Biochemical Pharmacology, 48(8).
DOI: 10.1016/0006-2952(94)90198-8
|















































































 GHS05;GHS06;GHS07;GHS08 Danger Details
GHS05;GHS06;GHS07;GHS08 Danger Details