Online Database of Chemicals from Around the World
| Hangzhou Hysen Pharma Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hysenpharma.cn | |||
 | +86 (571) 8829-8691 | |||
 | +86 (571) 2891-8299 | |||
 | sales@hysenpharma.cn | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Nanjing Finetech Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fine-chemtech.com | |||
 | +86 (25) 5207-8417 +86 17714198479 | |||
 | +86 (25) 5207-8417 | |||
 | sales@fine-chemtech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2007 | ||||
| BrightGene Bio-medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bright-gene.com | |||
 | +86 (512) 6255-1801 6255-1767 +86 13812696362 | |||
 | +86 (512) 6255-1799 | |||
 | kevinwan@bright-gene.com kevinwan0203@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2009 | ||||
| ZHIYU Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zhiyubiotech.com | |||
 | +86 (512) 6279-1916 | |||
 | +86 (512) 6279-1915 | |||
 | sales@zhiyubiotech.com info@zhiyubiotech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Renheng Pharm Limited | China | |||
|---|---|---|---|---|
 | www.renheng.com.cn | |||
 | +86 (20) 3998-1653 | |||
 | +86 (20) 3998-1158 | |||
 | sales@renheng.com.cn | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Tecoland Corporation | USA | |||
|---|---|---|---|---|
 | www.tecoland.com | |||
 | +1 (732) 603-9577 | |||
 | +1 (732) 906-1522 | |||
 | info@tecoland.com | |||
| Chemical distributor since 2001 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Shanghai Min-biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.min-biotech.com | |||
 | +86 15190045345 | |||
 | sales@min-biotech.com | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | API >> Other chemicals |
|---|---|
| Name | Ferinject |
| Synonyms | Ferric carboxymaltose; Injectafer; 4-[5-[3,4-dihydroxy-6-(hydroxymethyl)-5-[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxyoxan-2-yl]oxy-3,4-dihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-2,3,5,6-tetrahydroxyhexanoate iron(3+) oxygen(2-) hydroxide hydrate |
| Molecular Structure | 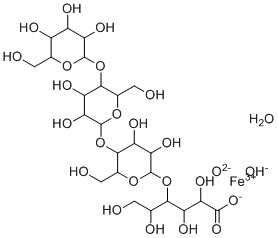 |
| Molecular Formula | C24H44FeO25- |
| Molecular Weight | 788.44 |
| CAS Registry Number | 9007-72-1 |
| EC Number | 813-933-0 |
| SMILES | C(C1C(C(C(C(O1)OC2C(OC(C(C2O)O)OC3C(OC(C(C3O)O)OC(C(CO)O)C(C(C(=O)[O-])O)O)CO)CO)O)O)O)O.O.[OH-].[O-2].[Fe+3] |
| Hazard Symbols |   GHS07;GHS08 Warning Details GHS07;GHS08 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335-H361-H373 Details | ||||||||||||||||||||||||
| Safety Statements | P203-P260-P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P318-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
|
Fehling injection, also known as Fehling's solution, was developed by German chemist Hermann von Fehling in the mid-19th century. Fehling originally devised the solution as a qualitative test for the presence of reducing sugars in analytical chemistry. Fehling injection is primarily used in laboratory settings as a reagent for detecting reducing sugars, such as glucose and fructose, in various chemical reactions. It consists of two separate solutions, Fehling A (aqueous copper(II) sulfate) and Fehling B (aqueous sodium potassium tartrate and sodium hydroxide), which are mixed in equal volumes before use. When heated with a solution containing reducing sugars, Fehling's solution undergoes a characteristic color change from blue to brick-red, indicating the presence of reducing sugars. References 2024. No longer to be ignored: Hypophosphatemia following intravenous iron administration. Reviews in Endocrine and Metabolic Disorders, 25(6). DOI: 10.1007/s11154-024-09926-5 2024. Randomised Controlled Trial Evaluating the Impact of Intravenous Iron (ferric carboxymaltose) Supplementation Among Epithelial Ovarian Cancer Patients with Anemia. Indian Journal of Surgical Oncology, 15(4). DOI: 10.1007/s13193-024-02149-9 2024. Efficacy of Intravenous Ferric Carboxymaltose in Heart Failure Patients with Iron Deficiency Anemia: A Meta-analysis of 6271 Patients. Clinical Drug Investigation, 44(11). DOI: 10.1007/s40261-024-01401-y |
| Market Analysis Reports |