Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Minakem S.A.S. | France | |||
|---|---|---|---|---|
 | www.minakem.com | |||
 | +33 (3) 2064-6830 | |||
 | +33 (3) 2064-3198 | |||
 | contact@minakem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Jiangsu Senxuan Pharmaceutical and Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.taixingpharm.com | |||
 | +86 (523) 8748-4267 | |||
 | +86 (523) 8748-5113 | |||
 | taixing@taixingpharm.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Changzhou Laiyinda Medical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lydpharm.com | |||
 | +86 (519) 8825-1268 | |||
 | +86 (519) 8825-1078 | |||
 | lyd@hi2000.com | |||
| Chemical manufacturer since 1995 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Ningbo Sialon Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.newsialon.com | |||
 | +86 (574) 2880-9708 2880-9768 | |||
 | +86 (574) 5670-8448 | |||
 | sales@newsialon.com roywang0086@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Xuchang Chenhe Bio-pharmaceutical Tech. Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.the-chem.com | |||
 | +86 (374) 579-3128 +86 15836530076 | |||
 | +86 (374) 579-3128 | |||
 | wxh5168@126.com wxh5168@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Eastar Chemical Corporation | USA | |||
|---|---|---|---|---|
 | www.eastarchem.com | |||
 | +1 800-898-2436 | |||
 | +1 (877) 898-2436 | |||
 | info@eastarchem.com | |||
| Chemical manufacturer since 1989 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Changzhou Jintan ChengEn Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chengenchem.com | |||
 | +86 (519) 8019-0155 +86 13961176135 +86 13775159780 | |||
 | +86 (519) 8019-0155 | |||
 | sales@chengenchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Ningbo Syntame Biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.syntame.com | |||
 | +86 (574) 2880-2872 | |||
 | sales@syntame.com | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Dalian Honkai Chemical Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.honkai.com | |||
 | +86 (411) 8479-1969 | |||
 | +86 (411) 8482-0706 | |||
 | sales@honkai.com | |||
| Chemical manufacturer since 2005 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Molekula Ltd | UK | |||
|---|---|---|---|---|
 | www.molekula.com | |||
 | +44 (1747) 831-066 | |||
 | +44 (1747) 831-199 | |||
 | info@molekula.com | |||
| Chemical manufacturer | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| AB Chem, Inc. | Canada | |||
|---|---|---|---|---|
 | www.abcheminc.com | |||
 | +1 (514) 685-8688 | |||
 | +1 (514) 685-8488 | |||
 | sales@abcheminc.com | |||
| Chemical manufacturer | ||||
| Watanabe Chemical Ind., Ltd. | Japan | |||
|---|---|---|---|---|
 | www.watanabechem.co.jp | |||
 | +81 (82) 231-0540 | |||
 | +81 (82) 231-1451 | |||
 | inquiry@watanabechem.co.jp | |||
| Chemical manufacturer | ||||
| Sinova Corporation | USA | |||
|---|---|---|---|---|
 | www.sinovainc.com | |||
 | +1 (301) 961-1525 | |||
 | +1 (240) 235-4288 | |||
 | sales@sinovainc.com | |||
| Chemical manufacturer | ||||
| OmegaChem Inc. | Canada | |||
|---|---|---|---|---|
 | www.omegachem.com | |||
 | +1 (418) 837-4444 (800) 661-6342 | |||
 | +1 (418) 837-5196 | |||
 | info@omegachem.com | |||
| Chemical distributor | ||||
| Chemicrea Inc. | Japan | |||
|---|---|---|---|---|
 | www.chemicrea.com | |||
 | +81 (3) 6214-5656 | |||
 | +81 (3) 6214-5659 | |||
 | finechemical@chemicrea.co.jp | |||
| Chemical manufacturer | ||||
| ECA International Corporation | USA | |||
|---|---|---|---|---|
 | www.ecacorporation.com | |||
 | +1 (847) 358-8178 | |||
 | +1 (847) 358-8179 | |||
 | order@ecacorporation.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Peptide |
|---|---|
| Name | (R)-(-)-4-Phenyl-2-oxazolidinone |
| Synonyms | (4R)-4-phenyl-1,3-oxazolidin-2-one |
| Molecular Structure | 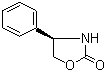 |
| Molecular Formula | C9H9NO2 |
| Molecular Weight | 163.17 |
| CAS Registry Number | 90319-52-1 |
| EC Number | 618-527-5 |
| SMILES | C1[C@H](NC(=O)O1)C2=CC=CC=C2 |
| Density | 1.2±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 131-133 °C (Expl.) |
| alpha | -55 ° (c=2, CHCl3) (Expl.) |
| Index of Refraction | 1.548, Calc.* |
| Boiling Point | 407.0±25.0 °C (760 mmHg), Calc.* |
| Flash Point | 200.0±23.2 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302+H312+H332 Details | ||||||||||||||||
| Safety Statements | P261-P264-P270-P271-P280-P301+P317-P302+P352-P304+P340-P317-P321-P330-P362+P364-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
(R)-(-)-4-Phenyl-2-oxazolidinone is a chiral organic compound belonging to the class of oxazolidinones, a family of compounds characterized by a five-membered ring containing one nitrogen and one oxygen atom. Specifically, this compound features a phenyl group attached to the 4-position of the oxazolidinone ring, with the (R)-(-) configuration indicating its chirality. The stereochemistry of this molecule is of significant importance, as the (R)-enantiomer often exhibits different chemical and biological activities compared to its (S)-counterpart. The discovery of (R)-(-)-4-phenyl-2-oxazolidinone can be attributed to ongoing efforts in synthetic chemistry to develop chiral compounds with specific properties, particularly in the realm of asymmetric synthesis. Oxazolidinones, due to their unique structure, have been the focus of various studies, particularly for their ability to serve as key intermediates in the synthesis of pharmaceuticals and biologically active compounds. The phenyl group attached to the oxazolidinone ring introduces an aromatic character that can significantly affect the compound’s solubility, stability, and reactivity, making it a valuable building block for further chemical modifications. One of the main applications of (R)-(-)-4-phenyl-2-oxazolidinone is in the pharmaceutical industry. The oxazolidinone structure is a core component of several important drug molecules, particularly in the development of antibiotics. For example, oxazolidinones are the basis for the class of drugs known as linezolid, which is used to treat multi-drug resistant bacterial infections. Although (R)-(-)-4-phenyl-2-oxazolidinone itself may not possess direct antibiotic activity, it serves as an important intermediate for the synthesis of more complex molecules that could have therapeutic effects. The ability to selectively synthesize one enantiomer, such as the (R)-form, is crucial for achieving the desired biological activity, as stereochemistry plays a key role in interactions with biological targets. In addition to its use in pharmaceuticals, (R)-(-)-4-phenyl-2-oxazolidinone also has applications in asymmetric synthesis, particularly in catalysis. The compound has been employed as a chiral ligand in various catalytic reactions, where its chiral environment can influence the stereochemistry of the resulting products. The presence of the phenyl group further contributes to the steric and electronic properties of the ligand, making it a useful tool for inducing chirality in a variety of chemical transformations. These properties make it valuable in the synthesis of fine chemicals, agrochemicals, and other specialty compounds. Moreover, (R)-(-)-4-phenyl-2-oxazolidinone has been studied for its potential applications in the field of materials science. The compound’s chiral nature and ability to interact with various other molecules suggest its use in designing chiral materials, such as liquid crystals or optoelectronic devices. The phenyl group’s ability to interact with other aromatic systems could also lead to interesting applications in molecular electronics and sensors. Overall, (R)-(-)-4-phenyl-2-oxazolidinone is a valuable compound in both synthetic and applied chemistry. Its importance as an intermediate for drug synthesis, as a chiral ligand in catalysis, and its potential uses in materials science highlight its versatility and relevance in various scientific fields. References 2009. Carbonylation with CO2 and Phosphorus Electrophiles: A Convenient Method for the Synthesis of 2-Oxazolidinones from 1,2-Amino Alcohols. Synlett, 2009(3). DOI: 10.1055/s-0028-1087531 2006. Design and Synthesis of a New Generation of ‘NH’-Ni(II) Complexes of Glycine Schiff Bases and their Unprecedented C-H vs. N-H Chemoselectivity in Alkyl Halide Alkylations and Michael Addition Reactions. Synlett, 2006(3). DOI: 10.1055/s-2006-926252 1997. Diastereoselective bromination of(R)-N-cinnamoyl-4-phenyloxazolidin-2-one in the presence of Lewis acids. Russian Chemical Bulletin, 46(5). DOI: 10.1007/bf02496131 |
| Market Analysis Reports |