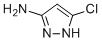
5-Chloro-1H-pyrazol-3-amine is a small nitrogen-containing heterocycle characterized by a pyrazole ring substituted with a chlorine atom at the 5-position and an amino group at the 3-position. The structure reflects the electron-rich nature of the pyrazole core, which is important in a wide range of chemical and biological contexts.
The discovery of 5-chloro-1H-pyrazol-3-amine is tied to the broader study of pyrazole chemistry, which has been a subject of intense investigation since the early 20th century. Researchers initially explored pyrazoles for their unique aromaticity and ability to serve as building blocks in various chemical reactions. The targeted introduction of halogens like chlorine into the pyrazole ring became a significant synthetic goal to modulate physical, chemical, and biological properties. 5-Chloro-1H-pyrazol-3-amine was obtained through methods such as selective chlorination of amino-substituted pyrazoles or by cyclization reactions involving appropriately substituted hydrazines and chlorinated precursors.
Applications of 5-chloro-1H-pyrazol-3-amine primarily revolve around its role as an intermediate in the synthesis of more complex molecules. In medicinal chemistry, pyrazole derivatives are widely recognized for their presence in numerous pharmacologically active compounds, including anti-inflammatory, analgesic, antiviral, and anticancer agents. The combination of an amino group and a chlorine atom on the pyrazole ring allows chemists to introduce this fragment into larger molecular frameworks, often to fine-tune the biological activity or improve pharmacokinetic profiles of lead compounds.
Specifically, 5-chloro-1H-pyrazol-3-amine can serve as a key starting material for the construction of kinase inhibitors, which target enzymes critical in the regulation of cellular activities. The presence of the chlorine atom can enhance binding affinity through hydrophobic interactions or influence the electronic distribution across the molecule, leading to improved selectivity and potency against specific biological targets.
In agricultural chemistry, derivatives of 5-chloro-1H-pyrazol-3-amine are explored for the development of herbicides and fungicides. The pyrazole ring system contributes to the inhibition of essential enzymes in plant pathogens or weeds, while chlorine substitution often improves environmental stability and bioavailability.
In material sciences, pyrazoles with functional groups like amino and chloro substituents are investigated for their utility in constructing coordination polymers and metal-organic frameworks (MOFs). The amino group can act as a coordinating site for metal ions, while the overall structure imparts interesting photophysical or electronic properties suitable for applications such as catalysis, sensing, and molecular storage.
The synthetic flexibility of 5-chloro-1H-pyrazol-3-amine is notable. The amino group enables subsequent reactions such as acylation, alkylation, or condensation with carbonyl compounds to form new heterocyclic systems. The chlorine atom offers a handle for cross-coupling reactions like Suzuki or Buchwald-Hartwig couplings, allowing for the introduction of diverse aryl or amine groups under palladium catalysis. These transformations extend the utility of 5-chloro-1H-pyrazol-3-amine far beyond its simple structure, making it a valuable tool in complex molecule synthesis.
Overall, 5-chloro-1H-pyrazol-3-amine exemplifies how strategic functionalization of a core heterocyclic system can produce versatile intermediates, supporting advances across medicinal, agricultural, and material chemistry disciplines.
|






 GHS07 Warning Details
GHS07 Warning Details