3-Chloro-1H-pyrazole is an organic compound with the molecular formula C3H2ClN2. It belongs to the pyrazole class of heterocyclic compounds, which are characterized by a five-membered ring structure containing two nitrogen atoms. The compound is specifically a halogenated pyrazole derivative, with a chlorine atom attached to the third position of the pyrazole ring.
The structure of 3-chloro-1H-pyrazole consists of a pyrazole ring where the nitrogen atoms are positioned at the 1 and 2 positions of the ring, and a chlorine atom is attached to the 3-position carbon. This substitution introduces a significant functional group that can modify the compound’s chemical reactivity, making it suitable for a variety of applications.
The synthesis of 3-chloro-1H-pyrazole can be achieved through several methods. One common approach involves the chlorination of pyrazole using chlorine gas or chlorinating agents such as phosphorus trichloride (PCl3). The reaction typically occurs under mild conditions and leads to the substitution of the chlorine atom at the 3-position. Another method involves direct halogenation using reagents such as N-chlorosuccinimide (NCS) or other electrophilic chlorinating agents.
3-Chloro-1H-pyrazole has a wide range of applications in various chemical and industrial fields. One of the most significant uses is in the synthesis of agrochemicals, including herbicides, fungicides, and insecticides. Pyrazole derivatives like 3-chloro-1H-pyrazole are often used as intermediates in the preparation of more complex molecules with biological activity. The chlorine substitution at the 3-position can enhance the stability and reactivity of the compound, making it an important building block for the development of crop protection chemicals.
In addition to its agricultural applications, 3-chloro-1H-pyrazole serves as an intermediate in pharmaceutical chemistry. Pyrazole derivatives have been studied for their biological activities, including antimicrobial, anti-inflammatory, and anticancer properties. The halogenated version of pyrazole, specifically 3-chloro-1H-pyrazole, can serve as a precursor for the synthesis of compounds with potential therapeutic effects. The presence of the chlorine atom can influence the compound's interactions with biological targets, making it an attractive structure for drug development.
Moreover, 3-chloro-1H-pyrazole is also utilized in materials science, particularly in the design of organic electronic materials and sensors. Pyrazole-based compounds have been investigated for their potential use in organic light-emitting diodes (OLEDs), organic solar cells, and other electronic devices. The chlorine substitution at the 3-position can influence the electronic properties of the molecule, which is useful in tuning the behavior of the material for specific applications.
The reactivity of 3-chloro-1H-pyrazole is influenced by the presence of the chlorine atom, which can participate in various nucleophilic substitution reactions. This makes it a valuable intermediate for the synthesis of other pyrazole derivatives or for further functionalization. For example, the chlorine atom can be replaced with other functional groups, such as amines or alkyl groups, to generate a variety of modified pyrazole compounds.
In summary, 3-chloro-1H-pyrazole is an important chemical intermediate used in the synthesis of agrochemicals, pharmaceuticals, and materials. Its structure, featuring a chlorine atom at the 3-position of the pyrazole ring, makes it reactive and versatile for a range of chemical transformations. Its applications span industries from agriculture to electronics, highlighting its significance in modern chemical synthesis. Further research into its reactivity and applications could lead to the development of more efficient and effective compounds in these fields.
|












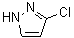
 GHS07 Warning Details
GHS07 Warning Details