Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Win-Win Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.win-winchemical.com | |||
 | +86 (577) 6449-8589 +86 15325081899 | |||
 | +86 (577) 5699-4596 | |||
 | sales@win-winchemical.com winwinchemical@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai CR Corporation Limited | China | |||
|---|---|---|---|---|
 | www.crcorporation.cn | |||
 | +86 (21) 5736-6208 +86 13917420128 | |||
 | +86 (21) 5736-6228 | |||
 | fred.wen@crcorporation.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Carbone Scientific Co., Ltd. | UK | |||
|---|---|---|---|---|
 | www.carbonesci.com | |||
 | +44 (870) 486-8629 | |||
 | +44 (870) 288-7399 | |||
 | sales@carbonesci.com | |||
| Chemical distributor | ||||
| Classification | Organic raw materials >> Nitrile compound |
|---|---|
| Name | 3-Fluoro-2-iodobenzonitrile |
| Molecular Structure | 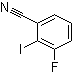 |
| Molecular Formula | C7H3FIN |
| Molecular Weight | 247.01 |
| CAS Registry Number | 916792-62-6 |
| EC Number | 811-190-7 |
| SMILES | C1=CC(=C(C(=C1)F)I)C#N |
| Solubility | Very slightly soluble (0.14 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.98±0.1 g/cm3 (20 °C 760 Torr), Calc.* |
| Boiling point | 275.5±25.0 °C (760 Torr), Calc.* |
| Flash point | 120.4±23.2 °C, Calc.* |
| Index of refraction | 1.63 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS06;GHS07 Danger Details GHS06;GHS07 Danger Details |
|---|---|
| Risk Statements | H301-H315-H319-H335 Details |
| Safety Statements | P261-P262-P264-P264+P265-P270-P271-P280-P301+P316-P302+P352-P304+P340-P305+P351+P338-P316-P319-P321-P330-P332+P317-P337+P317-P361+P364-P362+P364-P403+P233-P405-P501 Details |
| SDS | Available |
|
3-Fluoro-2-iodobenzonitrile is an aromatic compound featuring a benzene ring substituted with a fluorine atom at the 3-position, an iodine atom at the 2-position, and a nitrile group at the 1-position. This specific pattern of substitution combines the electronic effects of the electronegative fluorine and the bulky, reactive iodine with the electron-withdrawing character of the nitrile group, making the molecule particularly interesting for synthetic applications. The discovery of 3-fluoro-2-iodobenzonitrile is part of the broader exploration of halogenated benzonitriles, a class of compounds that became prominent as chemists sought versatile intermediates for the preparation of more complex organic molecules. The introduction of both a fluorine and an iodine atom onto a benzonitrile framework is typically achieved through carefully controlled halogenation reactions. In many cases, regioselective methods are employed, involving direct halogenation of fluorobenzonitriles or halogen exchange reactions starting from suitable precursors. The development of these methods was driven by the need for functionalized arenes that could participate in cross-coupling reactions, which have become central tools in organic synthesis. Applications of 3-fluoro-2-iodobenzonitrile are primarily linked to its use as a building block in the synthesis of pharmaceuticals, agrochemicals, and advanced materials. The iodine substituent makes the molecule particularly reactive in metal-catalyzed cross-coupling reactions, such as Suzuki-Miyaura, Sonogashira, or Heck reactions. These processes allow for the formation of new carbon-carbon or carbon-heteroatom bonds under mild conditions, making 3-fluoro-2-iodobenzonitrile a highly valuable synthon for constructing more complex structures. In pharmaceutical research, fluorinated arenes are highly prized due to the ability of the fluorine atom to influence molecular properties such as lipophilicity, metabolic stability, and binding affinity to biological targets. The presence of a nitrile group can further enhance binding interactions through hydrogen bonding or dipolar interactions, which are crucial in the design of enzyme inhibitors or receptor modulators. Thus, derivatives of 3-fluoro-2-iodobenzonitrile are often explored as intermediates in the preparation of compounds with potential therapeutic activities, including antitumor, antiviral, or anti-inflammatory agents. In the field of agrochemistry, compounds derived from 3-fluoro-2-iodobenzonitrile can be incorporated into the design of herbicides, fungicides, and insecticides. The structural framework of the molecule allows for the fine-tuning of biological activity by facilitating the introduction of various substituents at the 2-position via cross-coupling, while the nitrile group contributes to the overall bioactivity. In material sciences, fluorinated and iodinated aromatic compounds are utilized in the preparation of organic electronic materials, such as organic light-emitting diodes (OLEDs) and organic photovoltaics (OPVs). The unique combination of electronic properties imparted by the fluorine and iodine atoms can be exploited to engineer the energy levels of organic semiconductors, thereby improving their performance characteristics. Synthetically, 3-fluoro-2-iodobenzonitrile can undergo further functionalization through a variety of pathways. For example, the iodine atom can be replaced with aryl, vinyl, alkynyl, or amino groups using palladium-catalyzed reactions, enabling the rapid construction of diverse molecular architectures. The nitrile group can also be transformed into amides, carboxylic acids, or other functional groups through hydrolysis or other nucleophilic processes, providing additional avenues for derivatization. Overall, 3-fluoro-2-iodobenzonitrile serves as a valuable intermediate that bridges the gap between simple starting materials and complex target molecules across multiple domains of chemical research and industry. |
| Market Analysis Reports |