Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Boron Molecular Inc. | USA | |||
|---|---|---|---|---|
 | www.boronmolecular.com | |||
 | +1 (919) 981-4441 | |||
 | +1 (919) 981-4442 | |||
 | sales@boronmolecular-usa.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Win-Win Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.win-winchemical.com | |||
 | +86 (577) 6449-8589 +86 15325081899 | |||
 | +86 (577) 5699-4596 | |||
 | sales@win-winchemical.com winwinchemical@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Chiron AS | Norway | |||
|---|---|---|---|---|
 | www.chiron.no | |||
 | +47 (73) 874-490 | |||
 | +47 (73) 874-499 | |||
 | chiron@chiron.no | |||
| Chemical manufacturer | ||||
| Chem Service, Inc. | USA | |||
|---|---|---|---|---|
 | www.chemservice.com | |||
 | +1 (610) 692-3026 | |||
 | +1 (610) 692-8729 | |||
 | info@chemservice.com | |||
| Chemical manufacturer since 1962 | ||||
| Crescent Chemical Co. Inc. | USA | |||
|---|---|---|---|---|
 | www.crescentchemical.com | |||
 | +1 (631) 348-0333 | |||
 | +1 (631) 348-0913 | |||
 | crescent@creschem.com | |||
| Chemical distributor | ||||
| Frontier Scientific Services, Inc. | USA | |||
|---|---|---|---|---|
 | www.frontierssi.com | |||
 | +1 (302) 266-6891 (888) 577-2734 | |||
 | +1 (302) 266-8296 | |||
 | customerservice@frontierssi.com | |||
| Chemical manufacturer | ||||
| Chemtos | USA | |||
|---|---|---|---|---|
 | www.chemtos.com | |||
 | +1 (512) 686-4444 | |||
 | +1 (512) 858-4445 | |||
 | info@chemtos.com | |||
| Chemical manufacturer | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| Trigona oHG | Germany | |||
|---|---|---|---|---|
 | www.trigona.de | |||
 | +49 (611) 962-5283 | |||
 | +49 (611) 962-9032 | |||
 | info@trigona.de | |||
| Chemical manufacturer | ||||
| Spectrum Info Ltd. | Ukraine | |||
|---|---|---|---|---|
 | www.spec-info.com | |||
 | +38 (044) 573-5462 | |||
 | +38 (044) 573-2397 | |||
 | spec-info@spec-info.com | |||
| Chemical manufacturer | ||||
| Toronto Research Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.trc-canada.com | |||
 | +1 (416) 665-9696 | |||
 | +1 (416) 665-4439 | |||
 | info@trc-canada.com | |||
| Chemical manufacturer since 1982 | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic hydrocarbon reagent |
|---|---|
| Name | 4-Aminobiphenyl |
| Synonyms | 1,1'-Biphenyl-4-amine; 4-Biphenylylamine; 4-Phenylaniline |
| Molecular Structure | 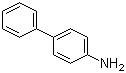 |
| Molecular Formula | C12H11N |
| Molecular Weight | 169.22 |
| CAS Registry Number | 92-67-1 |
| EC Number | 202-177-1 |
| SMILES | C1=CC=C(C=C1)C2=CC=C(C=C2)N |
| Solubility | Slightly soluble |
|---|---|
| Density | 1.16 g/mL |
| Melting point | 52-54 °C |
| Boiling point | 191 °C (15 mmHg) |
| Flash point | 113 °C |
| Hazard Symbols |   GHS07;GHS08 Danger Details GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H340-H341-H350 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P264-P270-P280-P301+P317-P318-P330-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 3077 | ||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||
|
4-Aminobiphenyl is an important organic compound used extensively in chemical synthesis and materials science. The compound, consisting of a biphenyl structure with an amino group attached at the 4-position of one of the phenyl rings, has found applications in various fields due to its unique chemical properties. The discovery of 4-Aminobiphenyl emerged from studies focused on the functionalization of biphenyl derivatives. Biphenyl, a compound featuring two connected phenyl rings, has been explored for its potential in a variety of chemical applications. The introduction of an amino group at the 4-position significantly alters the chemical behavior of biphenyl, opening up new avenues for its use. Synthesis of 4-Aminobiphenyl typically involves the nitration of biphenyl followed by a reduction step. The initial nitration introduces a nitro group at the desired position, which is subsequently reduced to an amino group. This method allows for precise control over the introduction of the amino group, which is crucial for the compound's applications. One of the primary applications of 4-Aminobiphenyl is in organic synthesis. It serves as a versatile building block in the preparation of various functionalized biphenyl derivatives. The amino group provides a site for further chemical modifications, enabling the synthesis of complex molecules. This property is particularly useful in the development of pharmaceuticals, where 4-Aminobiphenyl is used as a precursor in the synthesis of various therapeutic agents. In materials science, 4-Aminobiphenyl is employed in the development of organic electronic materials. The amino group can participate in polymerization reactions, leading to the formation of conducting polymers and other materials with desirable electronic properties. These materials are used in applications such as organic light-emitting diodes (OLEDs) and organic photovoltaic cells (OPVs), where their ability to conduct electricity and interact with light is critical. Another significant application of 4-Aminobiphenyl is in the field of dyes and pigments. The amino group can be utilized to synthesize various azo dyes, which are widely used in the textile industry. The presence of the amino group allows for the formation of azo bonds with other aromatic compounds, leading to the development of vibrant and stable dyes. 4-Aminobiphenyl has also been studied for its potential in medicinal chemistry. Its structure allows for the introduction of additional functional groups, making it a valuable intermediate in the synthesis of biologically active compounds. Research into the compound's biological activity has revealed its potential as a precursor for the development of new pharmaceuticals with specific therapeutic properties. In summary, 4-Aminobiphenyl is a versatile compound with significant applications in organic synthesis, materials science, and dye chemistry. Its ability to act as a building block for various chemical transformations highlights its importance in advancing both academic research and industrial applications. The ongoing study of 4-Aminobiphenyl continues to provide valuable insights into its potential uses and benefits. References 2005. Detection and Quantification of N-(Deoxyguanosin-8-yl)-4-aminobiphenyl Adducts in Human Pancreas Tissue Using Capillary Liquid Chromatography-Microelectrospray Mass Spectrometry. Chemical Research in Toxicology, 18(3). DOI: 10.1021/tx049692l 2005. High-performance liquid chromatography method for determination of N-glucuronidation of 4-aminobiphenyl by mouse, rat, and human liver microsomes. Analytical Biochemistry, 340(2). DOI: 10.1016/j.ab.2005.01.048 2014. Theoretical Studies of the Mechanism of N-Hydroxylation of Primary Aromatic Amines by Cytochrome P450 1A2: Radicaloid or Anionic?. Chemical Research in Toxicology, 27(1). DOI: 10.1021/tx400376u |
| Market Analysis Reports |