Online Database of Chemicals from Around the World
| Zhejiang Yangfan New Materials Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shoufu.com | |||
 | +86 (571) 8890-2510 8890-2504 8890-2511 | |||
 | +86 (571) 8890-2500 | |||
 | shoufu@shoufuchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Smartchem (Beijing) Ltd. | China | |||
|---|---|---|---|---|
 | www.smartchembeijing.com | |||
 | +86 (10) 5720-3829 5130-5129 +86 13301124250 | |||
 | +86 (10) 5130-5388 | |||
 | sales@smartchembeijing.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Chiron AS | Norway | |||
|---|---|---|---|---|
 | www.chiron.no | |||
 | +47 (73) 874-490 | |||
 | +47 (73) 874-499 | |||
 | chiron@chiron.no | |||
| Chemical manufacturer | ||||
| Eburon Organics bvba | Belgium | |||
|---|---|---|---|---|
 | www.eburon-organics.com | |||
 | +32 (51) 335-068 | |||
 | +32 (51) 335-064 | |||
 | sales@eburon-organics.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridine compound >> Methylpyridine |
|---|---|
| Name | Thianthrene |
| Molecular Structure | 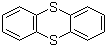 |
| Molecular Formula | C12H8S2 |
| Molecular Weight | 216.31 |
| CAS Registry Number | 92-85-3 |
| EC Number | 202-197-0 |
| SMILES | C1=CC=C2C(=C1)SC3=CC=CC=C3S2 |
| Melting point | 151-155 °C |
|---|---|
| Boiling point | 364-366 °C |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P305+P351+P338 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Thianthrene is a sulfur-containing heterocyclic compound that was first synthesized in the mid-19th century by treating diphenyl disulfide with sulfur monochloride. Its structure consists of two benzene rings fused to a central heterocycle containing two sulfur atoms. This unique arrangement was elucidated by structural studies in the early 20th century, and thianthrene became a noteworthy compound in organic chemistry for its aromatic stability and electronic properties. Thianthrene has a wide range of uses in the electronics industry, especially in organic electronics. Its planar structure and ability to delocalize π electrons on the sulfur atom and benzene ring make it an important component in organic semiconductors. It is used to develop materials for organic light-emitting diodes (OLEDs), organic photovoltaics (OPVs), and organic field-effect transistors (OFETs). These materials facilitate the production of flexible, lightweight, and efficient electronic devices. In synthetic organic chemistry, thianthrene is used as a reagent and intermediate. Its reactivity allows the introduction of sulfur atoms into organic molecules, aiding in the synthesis of sulfur-containing compounds. Thianthrene is capable of undergoing oxidation and reduction reactions and is therefore useful in a variety of chemical transformations, including the synthesis of thianthrene derivatives and related heterocycles. Thianthracene derivatives are used to produce dyes and pigments. Their electronic properties give the compounds intense and stable colors suitable for use in textiles, inks, and coatings. The sulfur atom in thianthracene enhances the lightfastness and stability of these dyes, making them valuable in creating long-lasting and vibrant colors. Thianthracene is used in the field of polymer science to develop advanced materials. Its incorporation into polymer backbones or as a comonomer helps produce polymers with enhanced thermal stability and electronic properties. These polymers can be used as high-performance materials in electronic devices, sensors, and other areas where stability and conductivity are required. In medicinal chemistry, thianthracene derivatives are intensively studied for their biological activities. Compounds containing the thianthracene core have shown potential as antimicrobial, anticancer, and anti-inflammatory agents. The sulfur atom in thianthracene helps it interact with biological targets, making it a promising scaffold in drug development. References 2022. Recent Advances in Thianthrenation/Phenoxathiination Enabled Site-Selective Functionalization of Arenes. Synthesis, 54(24). DOI: 10.1055/s-0041-1737493 2022. Alkylation Reactions with Alkylsulfonium Salts. Synthesis, 54(7). DOI: 10.1055/a-1677-5971 2023. A Comprehensive Study of the Structure and Conformational Behavior of 5-Phenylthianthrenium Triflate. Russian Journal of General Chemistry, 93(15). DOI: 10.1134/s1070363223150203 |
| Market Analysis Reports |