Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Beijing Huikang Boyuan Chemical Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.huikangchem.com | |||
 | +86 (10) 6886-2197 6886-7502 | |||
 | +86 (10) 6888-2204 | |||
 | market@huikangchem.com sales@huikangchem.com sales3@huikangchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Chengdu Hongsheng Sci-Tech Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cdchemical.com | |||
 | +86 (28) 3769-0186 +86 13568974161 | |||
 | +86 (28) 8587-6205 | |||
 | cdchemical@163.com cdchemi@gmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Infinity Scientific | China | |||
|---|---|---|---|---|
 | www.infsci.com | |||
 | +86 400-106-2016 +86 18500163824 | |||
 | biz2@infsci.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Honyan Pharmaceutical (wuhan) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hoyanmedicine.com | |||
 | +86 15832900195 | |||
 | +86 (27) 8233-9698 | |||
 | grace@whhoyan.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: 008615832900195 | |||
 | WhatsApp:008615832900195 | |||
| Chemical manufacturer since 2018 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Wuhan Ruichi Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | whrchem.com | |||
 | +86 13545065237 | |||
 | admin@whrchem.com | |||
 | WhatsApp:+8613545065237 | |||
| Chemical manufacturer since 2021 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Mahidhara Chemicals Private Limited | India | |||
|---|---|---|---|---|
 | www.mahidhara.com | |||
 | +91 8455 240445 | |||
 | mahidharamarketing1@gmail.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2023 | ||||
| FAR Chemical, Inc. | USA | |||
|---|---|---|---|---|
 | www.far-chemical.com | |||
 | +1 (321) 723-6160 | |||
 | +1 (321) 723-8753 | |||
 | info@far-chemical.com | |||
| Chemical manufacturer since 1983 | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Boulder Scientific Company | USA | |||
|---|---|---|---|---|
 | www.bouldersci.com | |||
 | +1 (970) 535-4494 | |||
 | +1 (970) 535-4584 | |||
 | sales@bouldersci.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organomagnesium |
|---|---|
| Name | Ethylmagnesium bromide |
| Molecular Structure | 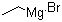 |
| Molecular Formula | C2H5BrMg |
| Molecular Weight | 133.27 |
| CAS Registry Number | 925-90-6 |
| EC Number | 213-127-3 |
| SMILES | C[CH2-].[Mg+2].[Br-] |
| Density | 1.02 g/mL (25 °C) (Expl.) |
|---|---|
| Flash point | -40 °C (closed cup) (Expl.) |
| Hazard Symbols |   GHS02;GHS05 Danger Details GHS02;GHS05 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H225-H260-H314 Details | ||||||||||||||||||||||||||||
| Safety Statements | P210-P223-P231+P232-P233-P240-P241-P242-P243-P260-P264-P280-P301+P330+P331-P302+P335+P334-P302+P361+P354-P303+P361+P353-P304+P340-P305+P354+P338-P316-P321-P363-P370+P378-P402+P404-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| Transport Information | UN 3399 | ||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Ethylmagnesium bromide is a widely utilized organometallic reagent classified as a Grignard reagent. It is a solution, typically in diethyl ether or tetrahydrofuran (THF), and plays a crucial role in organic synthesis. The reagent's discovery traces back to Victor Grignard in 1900, whose pioneering work with magnesium alkyl and aryl halides earned him the Nobel Prize in Chemistry in 1912. Ethylmagnesium bromide, like other Grignard reagents, has since become indispensable in the formation of carbon-carbon bonds and a variety of other chemical transformations. The preparation of ethylmagnesium bromide involves the reaction of ethyl bromide with magnesium metal in an anhydrous solvent like diethyl ether. The highly reactive nature of the magnesium center facilitates the generation of the ethylmagnesium bromide complex. The reaction is exothermic and requires careful handling due to the flammable nature of the reagents and the sensitivity of the Grignard reagent to moisture and air. In organic synthesis, ethylmagnesium bromide is employed as a nucleophile to add ethyl groups to electrophiles, including aldehydes, ketones, esters, and carbon dioxide. This reaction is fundamental in the synthesis of alcohols. For instance, the addition of ethylmagnesium bromide to formaldehyde yields ethanol, while its reaction with acetone produces 2-butanol. It is also utilized in the preparation of carboxylic acids through reaction with carbon dioxide and in the synthesis of alkanes via protonation. Ethylmagnesium bromide is also notable for its role in industrial chemistry. It is used to produce intermediates for pharmaceuticals, agrochemicals, and polymers. Its ability to form complex molecular frameworks makes it a cornerstone in the development of active pharmaceutical ingredients (APIs) and fine chemicals. Additionally, the reagent finds applications in organometallic coupling reactions, such as the Kumada coupling, which is pivotal in creating biaryl compounds. Despite its utility, ethylmagnesium bromide poses challenges due to its high reactivity. Handling requires an inert atmosphere, such as argon or nitrogen, to prevent degradation by oxygen or moisture. Advances in reagent formulation, such as stabilizing solutions and alternative solvents, continue to improve its usability and safety. Ethylmagnesium bromide exemplifies the power of Grignard chemistry in modern synthetic methodologies. Its ability to form carbon-carbon bonds and its versatility in diverse chemical transformations highlight its enduring importance in both academic research and industrial applications. References 2024. Synthesis of macrocyclic mono- and diolides based on new ω-hydroxyalkadienoic acids with (Z,Z)-1,5-diene moiety. Russian Chemical Bulletin, 73(6). DOI: 10.1007/s11172-024-4278-7 2024. Reactions of substituted 1-alkynylphosphines with Et2Zn catalyzed by the Cp2ZrCl2—EtMgBr system. Russian Chemical Bulletin, 73(7). DOI: 10.1007/s11172-024-4307-6 2023. Synthesis of macrocyclic and linear compounds with 1Z,5Z-diene and alkynylcarbinol fragments based on (5Z,9Z)-tetradeca-5,9-diene-1,14-diol. Russian Chemical Bulletin, 72(4). DOI: 10.1007/s11172-023-3855-1 |
| Market Analysis Reports |