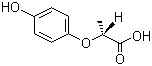
(R)-(+)-2-(4-Hydroxyphenoxy)propionic acid, also known as the (R)-enantiomer of fenofibric acid, is an important compound in medicinal chemistry. It originates from fenofibrate, a medication used to manage cholesterol levels and treat hyperlipidemia. The significance of this compound lies in its enhanced biological activity and potential applications across various fields.
The journey of (R)-(+)-2-(4-Hydroxyphenoxy)propionic acid begins with the development of fenofibrate in the 1970s. Fenofibrate is a prodrug that is metabolized into fenofibric acid, its active form, within the body. Among the two enantiomers of fenofibric acid, the (R)-enantiomer is notably more active. This is due to its higher affinity for peroxisome proliferator-activated receptor alpha (PPAR-α), a key regulator in lipid metabolism.
This compound is primarily used to treat dyslipidemia, a condition marked by abnormal lipid levels that increases the risk of cardiovascular diseases. Dyslipidemia often involves elevated triglyceride levels and low high-density lipoprotein (HDL) cholesterol. (R)-(+)-2-(4-Hydroxyphenoxy)propionic acid is effective in lowering triglycerides and raising HDL cholesterol. Clinical studies have shown that the (R)-enantiomer of fenofibric acid provides superior lipid-regulating effects compared to its counterpart, making it a valuable treatment for patients with metabolic syndrome and cardiovascular risks.
In addition to its medical uses, (R)-(+)-2-(4-Hydroxyphenoxy)propionic acid is significant in pharmacological research. It interacts with PPAR-α, offering insights into lipid metabolism and inflammation. Research into its mechanism of action is crucial for developing targeted treatments for lipid-related disorders and exploring new therapeutic options. The (R)-enantiomer's role in modulating PPAR-α pathways is central to understanding how selective modulators can impact metabolic processes.
Beyond medicine, (R)-(+)-2-(4-Hydroxyphenoxy)propionic acid is also valuable in industrial applications. Its chiral properties make it suitable for asymmetric synthesis, a process critical for producing enantiomerically pure compounds used in pharmaceuticals and agrochemicals. The compound’s stability and specificity facilitate the creation of high-purity products, highlighting its versatility and importance in chemical industries.
Overall, (R)-(+)-2-(4-Hydroxyphenoxy)propionic acid plays a crucial role in managing health conditions related to lipid metabolism, advancing pharmacological research, and contributing to industrial synthesis. Its continued study and application underscore its significance in both therapeutic and industrial contexts.
References
2024. Revealing the regulatory impact of nutrient on the production of (R)-2-(4-Hydroxyphenoxy)propanoic acid by Beauveria bassiana biofilms through comparative transcriptomics analyse. Bioprocess and Biosystems Engineering, 47(7).
DOI: 10.1007/s00449-024-03070-9
2020. Synthesizing Chiral Drug Intermediates by Biocatalysis. Applied Biochemistry and Biotechnology, 192(4).
DOI: 10.1007/s12010-020-03272-3
2020. Screening of Fungi Isolates for C-4 Hydroxylation of R-2-Phenoxypropionic Acid Based on a Novel 96-Well Microplate Assay Method. Applied Biochemistry and Biotechnology, 192(3).
DOI: 10.1007/s12010-020-03303-z
|

















































 GHS05 Danger Details
GHS05 Danger Details