Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Prisun Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.prisunpharma.com | |||
 | +86 (21) 5432-6183 | |||
 | prisun@prisunpharma.com | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Anhui Sholon New Material Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sholonchem.com | |||
 | +86 (0550) 526-1666 | |||
 | +86 (550) 526-1555 | |||
 | info@sholonchem.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Enamine Ltd. | Ukraine | |||
|---|---|---|---|---|
 | www.enamine.net | |||
 | +38 (044) 537-3218 | |||
 | +38 (044) 537-3253 | |||
 | enamine@enamine.net | |||
| Chemical manufacturer since 1991 | ||||
| Ukrorgsyntez Ltd. | Ukraine | |||
|---|---|---|---|---|
 | www.uorsy.com | |||
 | +38 (044) 531-9497 | |||
 | +38 (044) 531-9497 | |||
 | sales.eu@uorsy.com sales.us@uorsy.com | |||
| Chemical manufacturer since 2001 | ||||
| Ambinter | France | |||
|---|---|---|---|---|
 | www.ambinter.com | |||
 | +33 (1) 4524-4860 | |||
 | +33 (1) 4524-6241 | |||
 | contact@ambinter.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Aryl compounds |
|---|---|
| Name | 4-tert-Butoxystyrene |
| Synonyms | 1-(Tert-butoxy)-4-ethenylbenzene |
| Molecular Structure | 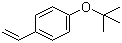 |
| Molecular Formula | C12H16O |
| Molecular Weight | 176.26 |
| CAS Registry Number | 95418-58-9 |
| EC Number | 619-127-3 |
| SMILES | CC(C)(C)OC1=CC=C(C=C1)C=C |
| Density | 0.9±0.1 g/cm3, Calc.*, 0.936 g/mL (Expl.) |
|---|---|
| Melting point | -38 °C (Expl.) |
| Index of Refraction | 1.523, Calc.*, 1.524 (Expl.) |
| Boiling Point | 256.8±9.0 °C (760 mmHg), Calc.*, 356.4-358.1 °C (Expl.) |
| Flash Point | 97.1±8.0 °C, Calc.*, 97 °C (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P305+P351+P338 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
4-tert-Butoxystyrene is an organic compound that is primarily used as a monomer in the production of polymers and copolymers. It is a derivative of styrene, where the hydrogen atom at the para-position of the benzene ring is replaced by a tert-butoxy group (–OC4H9). The tert-butoxy group provides steric hindrance, which can influence the reactivity and properties of the compound, particularly in polymerization reactions. The discovery of 4-tert-butoxystyrene can be traced to the broader investigation of substituted styrenes, which have been studied for their ability to undergo polymerization reactions. Substituted styrenes, including 4-tert-butoxystyrene, have attracted attention due to their ability to yield polymers with unique properties compared to unmodified styrene. The tert-butoxy group is particularly useful in controlling the polymerization process and in modifying the solubility, mechanical properties, and thermal stability of the resulting polymers. 4-tert-Butoxystyrene is commonly synthesized by the electrophilic aromatic substitution of styrene with tert-butyl alcohol in the presence of an acid catalyst, such as sulfuric acid. The reaction selectively introduces the tert-butoxy group at the para-position of the styrene ring. This synthesis route has been extensively studied and is well-established in organic chemistry. In addition, the use of tert-butyl peroxides as initiators in polymerization reactions is a typical method for producing polymers from 4-tert-butoxystyrene. One of the main applications of 4-tert-butoxystyrene is in the production of high-performance polymers. The presence of the bulky tert-butoxy group can impart desirable properties to the polymer, such as increased thermal stability, enhanced solubility in nonpolar solvents, and improved mechanical strength. Polymers made from 4-tert-butoxystyrene are often used in coatings, adhesives, and composite materials. These materials are useful in industries ranging from automotive to electronics, where high-performance materials with tailored properties are required. Another important application of 4-tert-butoxystyrene is in the synthesis of functional copolymers. By copolymerizing 4-tert-butoxystyrene with other monomers, such as styrene, methyl methacrylate, or acrylonitrile, it is possible to create copolymers with varying degrees of polarity, flexibility, and rigidity. These copolymers can be designed for specific applications, including in biomedical devices, packaging materials, and as advanced engineering plastics. Furthermore, 4-tert-butoxystyrene is also of interest in research applications, particularly in the fields of material science and polymer chemistry. The steric effects of the tert-butoxy group on polymerization kinetics and the structure of the resulting polymer chains have been subjects of study in academic research. These studies help scientists understand the relationship between monomer structure and polymer properties, aiding in the design of new materials for various applications. In summary, 4-tert-butoxystyrene is an important chemical used primarily in the production of specialized polymers and copolymers. Its discovery and development have contributed to the broader field of polymer chemistry, providing a means to tailor the properties of materials for specific industrial and research purposes. The compound's role in enhancing polymer stability and performance continues to be a valuable tool in both applied and academic settings. References 1990. Synthesis of block copolymers of p-methoxystyrene and vinyl ethers by the hydrogen iodide/zinc iodide initiating system. Polymer Bulletin, 24(2). DOI: 10.1007/bf00338494 2011. Micelle formation of polystyrene-block-poly(4-tert-butoxystyrene) in hexane. Colloid and Polymer Science, 289(15-16). DOI: 10.1007/s00396-011-2411-2 2019. Palladium/Copper Catalysis for Hydroarylation. Science of Synthesis. |
| Market Analysis Reports |