Online Database of Chemicals from Around the World
| Nanjing Finetech Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fine-chemtech.com | |||
 | +86 (25) 5207-8417 +86 17714198479 | |||
 | +86 (25) 5207-8417 | |||
 | sales@fine-chemtech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Yixing Xinyu Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xinyuchem.com | |||
 | +86 (510) 8744-6558 8744-1150 | |||
 | +86 (510) 8744-2808 / 8744-2345 | |||
 | sales@xinyuchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1993 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Jinan Zhongke Yitong Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jnzkyt.com | |||
 | +86 (531) 8865-8009 | |||
 | +86 (531) 8895-6078 | |||
 | 2871367557@qq.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Shanghai Valleyben Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.valleyben.com | |||
 | +86 13917011423 | |||
 | valleyben@qq.com | |||
| Chemical distributor since 2019 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Finetech Industry Limited | China | |||
|---|---|---|---|---|
 | www.finetechchem.com | |||
 | +86 (27) 8746-5837 +86 18971612321 | |||
 | +86 (27) 8777-2287 | |||
 | sales@finetechnology-ind.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Hangzhou Chuan Shuo Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.transol.cn | |||
 | +86 (571) 8760-9021 | |||
 | qian@transol.cn | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Suzhou Chenghe Pharmaceutical & Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chenghechem.com | |||
 | +86 (512) 5341-3000 8160-2999 | |||
 | +86 (512) 5341-0246 | |||
 | sales@chenghechem.com | |||
| Chemical manufacturer since 1995 | ||||
| Classification | Organic raw materials >> Amino compound >> Amide compound |
|---|---|
| Name | 4-Chloro-3-nitrobenzenesulfonamide |
| Molecular Structure | 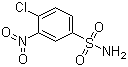 |
| Molecular Formula | C6H5ClN2O4S |
| Molecular Weight | 236.63 |
| CAS Registry Number | 97-09-6 |
| EC Number | 202-559-8 |
| SMILES | C1=CC(=C(C=C1S(=O)(=O)N)[N+](=O)[O-])Cl |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
4-Chloro-3-nitrobenzenesulfonamide is an aromatic sulfonamide compound featuring a benzene ring substituted with a chloro group (–Cl) at the 4-position, a nitro group (–NO2) at the 3-position, and a sulfonamide group (–SO2NH2) attached to the benzene ring. Its molecular structure combines electron-withdrawing substituents, which influence its chemical reactivity and physical properties. The compound is typically synthesized through the sulfonation of 4-chloro-3-nitroaniline or related intermediates, followed by conversion of the sulfonyl chloride intermediate to the sulfonamide via reaction with ammonia or amines. These steps require controlled conditions to achieve selective substitution and high purity, given the sensitivity of the nitro and chloro substituents to various reaction environments. 4-Chloro-3-nitrobenzenesulfonamide is primarily utilized as an intermediate in the synthesis of pharmaceuticals, agrochemicals, and dyes. The sulfonamide functionality is an important pharmacophore in medicinal chemistry, and the electron-withdrawing nitro and chloro groups modulate the compound's reactivity, enabling further chemical modifications such as reductions, substitutions, or coupling reactions. In pharmaceutical applications, derivatives of 4-chloro-3-nitrobenzenesulfonamide serve as precursors for drugs exhibiting antibacterial, anti-inflammatory, or diuretic activity. The compound’s reactivity also allows it to be employed in the preparation of sulfonamide-based enzyme inhibitors and other biologically active molecules. Analytical methods for characterizing 4-chloro-3-nitrobenzenesulfonamide include nuclear magnetic resonance (NMR) spectroscopy to confirm the substitution pattern on the aromatic ring and identify functional groups. Infrared (IR) spectroscopy is used to detect characteristic vibrations from the sulfonamide S=O stretches, nitro groups, and aromatic C–H bonds. Mass spectrometry (MS) and high-performance liquid chromatography (HPLC) provide molecular weight confirmation and purity analysis. Safety precautions when handling 4-chloro-3-nitrobenzenesulfonamide are consistent with those for aromatic sulfonamides and nitroaromatic compounds, which may pose toxicity or sensitization risks. Protective equipment such as gloves and eye protection, along with working in a well-ventilated environment, is recommended to minimize exposure. In summary, 4-chloro-3-nitrobenzenesulfonamide is a chemically versatile intermediate used in the synthesis of pharmaceuticals, agrochemicals, and dyes. Its well-defined synthetic routes, analytical characterization methods, and safety protocols support its effective and responsible application in industrial and research contexts. References 2023. The synthesis and study of carbonic anhydrase activity of sulfonamide-containing dibenzo[1,4]thiazepines. Chemistry of Heterocyclic Compounds, 59(12). DOI: 10.1007/s10593-024-03279-2 2022. Design, synthesis and anticancer activity studies of novel indole derivatives as Bcl-2/Mcl-1 dual inhibitors. Medicinal Chemistry Research, 31(12). DOI: 10.1007/s00044-022-02991-y 2011. Correlation analyses on binding affinity of substituted benzenesulfonamides with carbonic anhydrase using ab initio MO calculations on their complex structures (II). Bioorganic & Medicinal Chemistry Letters, 21(1). DOI: 10.1016/j.bmcl.2010.11.050 |
| Market Analysis Reports |