Tetraethylammonium fluoride hydrate (TEAF·xH2O) is a chemical compound consisting of tetraethylammonium cations (C2H5)4N+ and fluoride anions (F-) associated with water molecules. The compound is typically prepared by reacting tetraethylammonium salts, such as tetraethylammonium bromide or tetraethylammonium chloride, with sodium fluoride in the presence of water. This reaction leads to the formation of tetraethylammonium fluoride along with water of crystallization, resulting in a hydrate form.
The structure of tetraethylammonium fluoride hydrate involves the tetraethylammonium cations interacting with fluoride ions, which are often bound to water molecules in the crystalline structure. The degree of hydration can vary, leading to different forms of the compound depending on the conditions under which it is synthesized. This compound is known for its strong ionic nature and is primarily of interest due to its properties as a source of fluoride ions.
Tetraethylammonium fluoride hydrate has several applications, particularly in organic chemistry and materials science. One of its primary uses is as a source of fluoride ions in organic synthesis. Fluoride ions are highly nucleophilic and can be used to facilitate a range of chemical reactions, including nucleophilic substitutions and the preparation of fluorinated organic compounds. Tetraethylammonium fluoride hydrate serves as a safer and more easily handled source of fluoride compared to anhydrous fluoride salts, which are often highly reactive and difficult to store and manipulate.
In addition to its use in organic synthesis, tetraethylammonium fluoride hydrate is used in the preparation of ionic liquids. Ionic liquids are salts that are liquid at or near room temperature and are widely used as solvents in green chemistry and various industrial processes. The fluoride anion, in combination with tetraethylammonium cations, is used to produce ionic liquids that are stable, non-volatile, and useful in applications such as catalysis, electrochemical systems, and solvent extraction.
Tetraethylammonium fluoride hydrate also finds utility in the synthesis of other specialized materials. For example, it has been employed in the preparation of fluoride-containing metal-organic frameworks (MOFs) and in the modification of materials for electronic and catalytic applications. The compound’s ability to release fluoride ions in a controlled manner makes it valuable for these types of synthetic processes.
The compound is used in electrochemical applications as well, particularly in the development of fluoride-based ionic liquids and electrolytes for energy storage devices, such as batteries and capacitors. The strong ionic conductivity and stability of tetraethylammonium fluoride hydrate make it a useful component in these applications, contributing to the efficiency and performance of electrochemical systems.
Like other fluoride-containing compounds, tetraethylammonium fluoride hydrate must be handled with care. Fluoride ions are highly reactive and can be corrosive, making the compound potentially hazardous if not managed properly. Proper safety precautions, such as the use of gloves, goggles, and appropriate laboratory equipment, should be observed when working with this substance.
In conclusion, tetraethylammonium fluoride hydrate is a versatile chemical compound with applications in organic synthesis, the preparation of ionic liquids, and materials science. Its ability to release fluoride ions in a controlled manner makes it valuable in a variety of chemical processes, and its use in electrochemical and energy storage systems underscores its importance in modern chemical research and industrial applications.
References
1985 Crystal structure of tetraethylammonium fluoride-water (4/11), 4(C2H5)4N+F− · 11H2O, a clathrate hydrate containing linear chains of edge-sharing (H2O)4F− tetrahedra and bridging water molecules. Journal of Inclusion Phenomena and Macrocyclic Chemistry, 1, 55735.
DOI: 10.1007/bf00655735
2016 Process Chemistry in Antiviral Research. Topics in Current Chemistry, 1, 76.
DOI: 10.1007/s41061-016-0076-5
|













































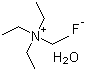
 GHS07 Warning Details
GHS07 Warning Details