Online Database of Chemicals from Around the World
| Shanghai B&C Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bcpharma.com | |||
 | +86 (21) 6597-9781 6597-9782 6597-9783 | |||
 | +86 (21) 6597-9780 | |||
 | info@greenchemintl.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Sichuan Zhongbang Technical Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zhongbangst.com | |||
 | +86 (830) 258-5019 +86 18982761636 | |||
 | +86 (830) 258-9033 | |||
 | sales@zhongbangst.com sheldon0chen@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Qyubiotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qyubiotech.com | |||
 | +86 (021) 3759-6619 | |||
 | +86 (021) 3759-6619 | |||
 | lioner@qyubiotech.com | |||
 | QQ Chat | |||
| Chemical distributor since 2015 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Alcohol |
|---|---|
| Name | 1,3,5-Adamantanetriol |
| Synonyms | adamantane-1,3,5-triol |
| Molecular Structure | 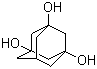 |
| Molecular Formula | C10H16O3 |
| Molecular Weight | 184.23 |
| CAS Registry Number | 99181-50-7 |
| SMILES | C1C2CC3(CC1(CC(C2)(C3)O)O)O |
| Density | 1.6±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.749, Calc.* |
| Boiling Point | 339.7±37.0 °C (760 mmHg), Calc.* |
| Flash Point | 168.7±21.1 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
1,3,5-adamantantriol is a unique compound belonging to the adamantane family that was first synthesized and identified in the mid-20th century. The discovery of this compound stems from the study of adamantane derivatives, which are known for their rigidity and cage-like structure. 1,3,5-adamantantriol is characterized by the presence of three hydroxyl (-OH) groups at positions 1, 3, and 5 of the adamantane backbone. This structural arrangement confers unique chemical properties that have found applications in various fields of science and industry. One notable application of 1,3,5-adamantantriol is its antiviral and antibacterial properties. Studies have shown that adamantane derivatives, including 1,3,5-adamantantriol, have inhibitory effects against certain viruses and bacteria. This makes it a potential candidate for the development of new antiviral and antibacterial agents, particularly against influenza viruses and other pathogens. In pharmaceutical research, 1,3,5-adamantantriol has been intensively studied for its role in drug delivery systems. Its rigidity and symmetrical structure can be used to create drug carriers or nanocarriers that are able to encapsulate therapeutic agents and deliver them to specific targets in the body. This application enhances drug stability, improves bioavailability and enables controlled release, which is valuable for improving the efficacy of drug treatments. The unique molecular structure of 1,3,5-adamantantriol also makes it suitable for materials science. It has been studied for polymer synthesis and as a building block for creating new materials with customized properties. Incorporating adamantane derivatives into polymers can enhance mechanical strength, thermal stability and chemical resistance, thereby expanding its potential applications in industries such as aerospace, automotive and electronics. 1,3,5-adamantantriol is a versatile organic synthetic chemical intermediate. Its hydroxyl groups can undergo a variety of chemical modifications, enabling the synthesis of complex organic molecules and drug intermediates. This versatility is valuable to researchers and chemists seeking to develop new compounds with specific functions and applications. In addition to antiviral and drug delivery applications, 1,3,5-adamantantriol has been studied in biomedical research for its potential therapeutic benefits. Research focuses on its interactions with biological systems and its ability to modulate cellular processes, which could lead to advances in the treatment of diseases such as cancer and neurodegenerative diseases. References 2020. Synthesis of 3,5-Bis(hydroxymethyl)adamantan-1-ols and 3,5-Bis(nitrooxymethyl)adamantan-1-yl Nitrates. Russian Journal of Organic Chemistry, 56(9). DOI: 10.1134/s1070428020090109 2017. Oxidative functionalization of adamantanes (review). Petroleum Chemistry, 57(2). DOI: 10.1134/s0965544117020128 2015. Improved approach towards synthesis of adamantane-1,3,5-triol. Russian Journal of General Chemistry, 85(8). DOI: 10.1134/s1070363215080071 |
| Market Analysis Reports |