Online Database of Chemicals from Around the World
| Fenhe Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fenhechem.com | |||
 | +86 (021) 3392-6068 | |||
 | julius.wei@fenhechem.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Pyrimidine |
|---|---|
| Name | Ethyl 5-amino-2-methylnicotinate |
| Molecular Structure | 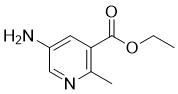 |
| Molecular Formula | C9H12N2O2 |
| Molecular Weight | 180.20 |
| CAS Registry Number | 60390-42-3 |
| SMILES | CCOC(=O)C1=C(N=CC(=C1)N)C |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302 Details |
| Safety Statements | P280-P305+P351+P338 Details |
| SDS | Available |
|
Ethyl 5-amino-2-methylnicotinate is a significant chemical compound with a range of applications in medicinal chemistry and organic synthesis. This molecule features a nicotinate core, which is a derivative of nicotinic acid, modified with an ethyl ester, an amino group, and a methyl substituent. The discovery of Ethyl 5-amino-2-methylnicotinate is rooted in efforts to develop functionalized nicotinate derivatives. Nicotinate derivatives are known for their diverse biological activities and utility in medicinal chemistry. The ethyl ester group at position 2 enhances the compound's solubility and reactivity, while the amino group at position 5 and the methyl group at position 2 add further complexity to its chemical properties. In medicinal chemistry, Ethyl 5-amino-2-methylnicotinate is of particular interest due to its potential therapeutic properties. The nicotinate core is associated with various bioactive compounds, and the specific modifications in this molecule can influence its interaction with biological targets. Researchers are exploring how the amino and methyl groups affect the compound’s pharmacological activity, potentially leading to new drug candidates. The presence of the amino group may enhance the molecule’s ability to interact with receptors or enzymes, making it a candidate for further development in drug discovery. The compound is also valuable in organic synthesis as an intermediate for creating more complex molecules. The ethyl ester and amino groups enable it to participate in a variety of chemical reactions, facilitating the synthesis of novel compounds with potential applications in pharmaceuticals and materials science. This versatility makes Ethyl 5-amino-2-methylnicotinate a useful reagent for chemists developing new synthetic methodologies. Furthermore, Ethyl 5-amino-2-methylnicotinate can be employed in the study of structure-activity relationships (SAR) in drug design. By analyzing how variations in the substituents affect biological activity, researchers can gain insights into designing more effective therapeutic agents. This compound serves as a valuable tool in understanding how functional groups influence the biological properties of nicotinate derivatives. In summary, Ethyl 5-amino-2-methylnicotinate is a compound with significant potential due to its unique structure and reactivity. Its applications in medicinal chemistry, organic synthesis, and SAR studies highlight its importance in advancing research and development across various scientific fields. References none |
| Market Analysis Reports |