Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Weiyel Inc | China | |||
|---|---|---|---|---|
 | www.weiyel.com | |||
 | +44 7421898692 | |||
 | ivy@weiyelbncc.com | |||
 | QQ Chat | |||
 | WeChat: +8615672728568 | |||
 | WhatsApp:+44 7421898692 | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Chem Service, Inc. | USA | |||
|---|---|---|---|---|
 | www.chemservice.com | |||
 | +1 (610) 692-3026 | |||
 | +1 (610) 692-8729 | |||
 | info@chemservice.com | |||
| Chemical manufacturer since 1962 | ||||
| Alfa Aesar. | USA | |||
|---|---|---|---|---|
 | www.alfa.com | |||
 | +1 (978) 521-6300 | |||
 | +1 (978) 521-6350 | |||
 | info@alfa.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Aldehyde |
|---|---|
| Name | Acetal |
| Synonyms | 1,1-Diethoxyacetal; 1,1-Diethoxyethane; Acetaldehyde diethyl acetal |
| Molecular Structure | 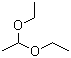 |
| Molecular Formula | C6H14O2 |
| Molecular Weight | 118.17 |
| CAS Registry Number | 105-57-7 |
| EC Number | 203-310-6 |
| FEMA | 2002 |
| SMILES | CCOC(C)OCC |
| Density | 0.825 |
|---|---|
| Melting point | -100 °C |
| Boiling point | 103 °C |
| Refractive index | 1.379-1.385 |
| Flash point | -21 °C |
| Water solubility | 46 g/L (25 °C) |
| Hazard Symbols |   GHS02;GHS07 Danger Details GHS02;GHS07 Danger Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H225-H315-H319 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P264-P264+P265-P280-P302+P352-P303+P361+P353-P305+P351+P338-P321-P332+P317-P337+P317-P362+P364-P370+P378-P403+P235-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| Transport Information | UN 1088 | ||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
Acetal, an organic compound type characterized by the general formula R₂C(OR')₂, is central in organic chemistry and industrial applications. Acetals were first studied in the 19th century as chemists began investigating compounds capable of protecting functional groups in complex synthesis processes. The term "acetal" commonly refers to any compound where two alkoxy groups are bonded to a single carbon atom, stemming from initial studies on acetaldehyde derivatives. The reactivity and stability of acetal linkages in both synthetic and biological systems have established acetals as valuable compounds in multiple fields. Structurally, acetals contain a carbon atom connected to two ether (–OR) groups and one or two alkyl (R) or aryl (Ar) groups. This configuration makes acetals highly stable under neutral or basic conditions, although they readily decompose in acidic environments. Acetals can be synthesized through the acid-catalyzed reaction of an aldehyde or ketone with an alcohol, forming a hemiacetal intermediate, which subsequently reacts with another alcohol molecule to yield the final acetal structure. This reactivity has made acetals valuable as protective groups in organic synthesis, shielding sensitive carbonyl groups during multi-step reactions. In industrial and commercial contexts, acetals play critical roles in materials and manufacturing. Polyoxymethylene (POM), also known as acetal resin, is a widely used thermoplastic derived from formaldehyde, with applications spanning automotive components, consumer electronics, and precision parts. Due to its high strength, low friction, and excellent chemical resistance, acetal resin is used in manufacturing gears, fasteners, and bearing surfaces. In food packaging and medical devices, acetals are valued for their stability and low moisture absorption, which ensure the longevity and safety of end products. Beyond manufacturing, acetals also serve in perfumery and pharmaceuticals, where they are synthesized to encapsulate and release fragrances and medicinal compounds gradually. Acetals derived from natural or synthetic aldehydes and alcohols are also used as flavoring agents due to their stability and pleasant aromas, contributing to the sensory qualities of food and beverages. Their stability and relatively low toxicity make them suitable for various consumer products, though regulatory standards govern their use, particularly in consumables. The discovery and utilization of acetals have made a lasting impact across chemistry and industry. Their protective qualities in synthetic chemistry, resilience in materials, and contribution to consumer products reflect the versatility and ongoing relevance of acetal compounds in modern applications. References 2024. Preparative scalable method for the synthesis of cyclic and acyclic acetals of chloropropiolaldehyde and their transformation into acetals of lithiumpropiolaldehyde. Russian Chemical Bulletin, 73(10), 2607-2614. DOI: 10.1007/s11172-024-4428-y 2013. In(III)/PhCO2H Binary Acid Catalyzed Tandem [2 + 2] Cycloaddition and Nazarov Reaction between Alkynes and Acetals. Organic Letters, 15(16), 4182-4185. DOI: 10.1021/ol4020464 2010. Direct Aerobic Photo-Oxidative Synthesis of Aromatic Methyl Esters from Methyl Aromatics via Dimethyl Acetals. Organic Letters, 12(15), 3494-3497. DOI: 10.1021/ol1014575 |
| Market Analysis Reports |