Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| The Dow Chemical Company | USA | |||
|---|---|---|---|---|
 | www.dow.com | |||
 | +1 (989) 633-1706 | |||
 | fmdcigs@dow.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Qingdao ZKHT Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zkhtchem.com | |||
 | +86 (532) 8689-8076 +86 15153292017 | |||
 | +86 (532) 8689-8076 | |||
 | zkhtchem@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Anhui Royal Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.royal-chem.com | |||
 | +86 (25) 8663-9253 | |||
 | +86 (25) 5227-9193 | |||
 | zoey.zhao@royal-chem.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Finetech Industry Limited | China | |||
|---|---|---|---|---|
 | www.finetechchem.com | |||
 | +86 (27) 8746-5837 +86 18971612321 | |||
 | +86 (27) 8777-2287 | |||
 | sales@finetechnology-ind.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Richman Chemical Inc. | USA | |||
|---|---|---|---|---|
 | www.richmanchemical.com | |||
 | +1 (215) 628-2946 | |||
 | +1 (215) 628-4262 | |||
 | kac@richmanchemical.com | |||
| Chemical manufacturer since 1988 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Sinocure Chemical Group | China | |||
|---|---|---|---|---|
 | www.sinocurechem.com | |||
 | +86 15550440621 | |||
 | info@sinocurechem.com | |||
| Chemical manufacturer since 2020 | ||||
| Scientific Polymer Products, Inc. | USA | |||
|---|---|---|---|---|
 | www.scientificpolymer.com | |||
 | +1 (585) 265-0413 | |||
 | +1 (585) 265-1390 | |||
 | custserv@scipoly.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Polycyclic compound |
|---|---|
| Name | Glycidyl methacrylate |
| Synonyms | 2,3-Epoxypropyl methacrylate; Methacrylic acid 2,3-epoxypropyl ester |
| Molecular Structure | 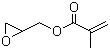 |
| Molecular Formula | C7H10O3 |
| Molecular Weight | 142.15 |
| CAS Registry Number | 106-91-2 |
| EC Number | 203-441-9 |
| SMILES | CC(=C)C(=O)OCC1CO1 |
| Density | 1.042 |
|---|---|
| Boiling point | 189 °C |
| Refractive index | 1.449-1.451 |
| Flash point | 76 °C |
| Water solubility | 0.5-1.0 g/100 mL at 20 °C |
| Hazard Symbols |     GHS05;GHS06;GHS07;GHS08 Danger Details GHS05;GHS06;GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302+H332-H302-H311-H312-H314-H315-H317-H318-H319-H332-H335-H341-H350-H360-H370-H372 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P260-P261-P262-P264-P264+P265-P270-P271-P272-P280-P301+P317-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P351+P338-P305+P354+P338-P308+P316-P316-P317-P318-P319-P321-P330-P332+P317-P333+P317-P337+P317-P361+P364-P362+P364-P363-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2810 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Glycidyl methacrylate (GMA) is a versatile chemical compound with the molecular formula C₇H₁₀O₃, characterized by the presence of both an epoxide group and a methacrylate group. This dual functionality makes GMA a valuable monomer in various polymerization reactions, leading to its widespread use in the production of specialty polymers, coatings, adhesives, and composites. The discovery of glycidyl methacrylate can be traced back to the mid-20th century when researchers were exploring new monomers for use in polymer chemistry. The compound was identified as particularly promising due to its reactive epoxide group, which allows it to undergo copolymerization with a wide range of other monomers, and its methacrylate group, which provides a site for radical polymerization. This unique combination of properties has made GMA an important building block in the synthesis of functional polymers. One of the primary applications of glycidyl methacrylate is in the production of epoxy resins. When GMA is copolymerized with other monomers, it forms polymers with reactive epoxide groups that can be further cross-linked using curing agents. These epoxy-based polymers are widely used in the coatings industry, particularly for protective coatings on metals and other surfaces. The high reactivity of the epoxide groups in GMA-based polymers allows for strong adhesion to substrates and excellent resistance to chemicals and environmental factors. Glycidyl methacrylate is also used in the production of acrylic resins, which are employed in a variety of applications, including adhesives, sealants, and composites. The presence of the methacrylate group in GMA enables its copolymerization with other acrylic monomers, resulting in resins with enhanced mechanical properties and durability. These acrylic resins are often used in the automotive and aerospace industries, where they provide lightweight, high-performance materials for coatings, structural components, and adhesives. In addition to its use in coatings and adhesives, glycidyl methacrylate is utilized in the synthesis of ion exchange resins, which are important in water treatment and purification processes. The reactive epoxide group in GMA allows for the introduction of functional groups that can selectively bind to ions, making these resins effective in removing contaminants from water. This application highlights the versatility of GMA in creating polymers with specific and tailored functionalities. Furthermore, GMA is employed in the development of biomedical materials, particularly in the fabrication of hydrogels and drug delivery systems. The ability of GMA to undergo copolymerization with hydrophilic monomers allows for the creation of hydrogels with controlled swelling properties and biocompatibility. These materials are used in wound dressings, tissue engineering, and as carriers for controlled drug release. The handling of glycidyl methacrylate requires careful attention to safety, as the compound is known to be a skin sensitizer and can cause irritation upon contact. Proper protective equipment and handling procedures are essential when working with GMA to minimize exposure and potential health risks. Overall, the discovery and application of glycidyl methacrylate have had a significant impact on the field of polymer chemistry, enabling the development of a wide range of high-performance materials with applications across numerous industries. References 2014. Preparation of a novel weak cation exchange/hydrophobic interaction chromatography dual-function polymer-based stationary phase for protein separation using “thiol-ene click chemistry”. Analytical and Bioanalytical Chemistry, 407(6), 1729-1740. DOI: 10.1007/s00216-014-8400-9 2012. Equilibrium and kinetics study on hexavalent chromium adsorption onto diethylene triamine grafted glycidyl methacrylate based copolymers. Journal of Hazardous Materials, 217-218, 315-322. DOI: 10.1016/j.jhazmat.2011.12.079 2007. Living Radical Polymerizations with Germanium, Tin, and Phosphorus Catalysts − Reversible Chain Transfer Catalyzed Polymerizations (RTCPs). Journal of the American Chemical Society, 129(42), 12628-12629. DOI: 10.1021/ja0755820 |
| Market Analysis Reports |