Online Database of Chemicals from Around the World
| Changzhou Huaren Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.huarenchem.com | |||
 | +86 (519) 8812-2965 | |||
 | +86 (519) 8810-4456 | |||
 | huarenchem@vip.163.com sales@huarenchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2000 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| HBCChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.hbcchem-inc.com | |||
 | +1 (510) 219-6317 | |||
 | +1 (510) 675-0318 | |||
 | sales@hbcchem-inc.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Witofly Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.witofly.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales@witofly.com | |||
 | QQ Chat | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Kessie Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kessiechem.com | |||
 | +86 (519) 8698-7916 +86 15851999766 | |||
 | +86 (519) 8690-8212 | |||
 | info@kessiechem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Jiangsu Nengyue New Material Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jiangsunengyue.com | |||
 | +86 13376001577 | |||
 | robin@jiangsunengyue.com | |||
 | WhatsApp:+8613376001577 | |||
| Chemical manufacturer since 2022 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Ether |
|---|---|
| Name | (R)-Glycidyl butyrate |
| Synonyms | Oxiran-2-ylmethyl butyrate |
| Molecular Structure | 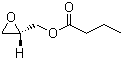 |
| Molecular Formula | C7H12O3 |
| Molecular Weight | 144.17 |
| CAS Registry Number | 60456-26-0 |
| EC Number | 611-977-3 |
| SMILES | CCCC(=O)OC[C@H]1CO1 |
| Density | 1.035 |
|---|---|
| Boiling point | 192.6 °C |
| Refractive index | 1.427-1.429 |
| Flash point | 90 °C |
| alpha | -30 ° (c=neat) |
| Water solubility | insoluble |
| Hazard Symbols |   GHS07;GHS08 Danger Details GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H334-H335 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P233-P260-P261-P264-P264+P265-P270-P271-P280-P284-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P342+P316-P362+P364-P403-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
(R)-Glycidyl butyrate is an enantiomerically pure compound that plays a crucial role as an intermediate in organic synthesis, particularly in the preparation of chiral molecules. This compound belongs to the class of glycidyl esters, characterized by the presence of an epoxide functional group, which is highly reactive and useful in various chemical reactions. The (R)-configuration of glycidyl butyrate refers to its specific stereochemistry, which is important for applications where chirality is required to produce enantiomerically pure products. The discovery of (R)-glycidyl butyrate can be traced back to advances in asymmetric synthesis, a field dedicated to producing molecules with specific three-dimensional arrangements. The development of efficient methods to synthesize enantiomerically pure epoxides like (R)-glycidyl butyrate has been driven by the increasing demand for chiral intermediates in the pharmaceutical, agrochemical, and fine chemical industries. The ability to control the stereochemistry of such compounds is critical in ensuring the efficacy and safety of biologically active molecules. (R)-Glycidyl butyrate has a structure consisting of an epoxide ring attached to a butyrate ester group. The reactivity of the epoxide group makes this compound valuable for a wide range of chemical transformations. One of its primary uses is in the synthesis of chiral alcohols and diols, which are important intermediates in the production of pharmaceuticals. For example, (R)-glycidyl butyrate can undergo ring-opening reactions with nucleophiles, leading to the formation of chiral alcohols that are used in the synthesis of active pharmaceutical ingredients (APIs). The stereochemistry of the epoxide ensures that the resulting products have the desired three-dimensional structure, which is often crucial for the biological activity of the final compound. In addition to its application in pharmaceutical synthesis, (R)-glycidyl butyrate is also used in the production of fine chemicals and materials. It can be incorporated into polymers, where the presence of the epoxide group allows for cross-linking and other modifications that enhance the properties of the material. Chiral glycidyl esters like (R)-glycidyl butyrate are particularly valuable in the production of optically active polymers, which have applications in areas such as liquid crystal displays (LCDs) and other advanced optical technologies. Research has also explored the use of (R)-glycidyl butyrate in the field of asymmetric catalysis. Its highly reactive epoxide group can be used to create chiral environments that promote enantioselective reactions, enabling the synthesis of complex molecules with high stereochemical fidelity. This application is particularly relevant in the development of new drugs and agrochemicals, where the precise control of stereochemistry can significantly impact the effectiveness and safety of the final product. The safety and environmental impact of (R)-glycidyl butyrate must also be considered, as the reactive nature of the epoxide group can pose certain hazards. Proper handling and storage procedures are necessary to prevent unwanted reactions and ensure safe use in industrial and laboratory settings. Moreover, efforts are being made to develop more sustainable methods for its synthesis, with a focus on reducing waste and improving the environmental profile of the production processes. References 2018. Comparative features between recombinant lipases CALA-like from U. maydis and CALA from C. antarctica in thermal stability and selectivity. Biotechnology Letters, 40(12). DOI: 10.1007/s10529-018-2630-4 2008. Resolution of (R,S)-(+/-)-glycidyl butyrate with immobilized Y-11 Trichosporon capitatum lipase in gelatin-containing microemulsion-based organogels. Colloids and surfaces. B, Biointerfaces, 66(2). DOI: 10.1016/j.colsurfb.2008.08.011 2015. Enantioselective Kinetic Resolution of Esters of Chiral Primary Alcohols by Porcine Pancreatic Lipase. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-214-00121 |
| Market Analysis Reports |