Online Database of Chemicals from Around the World
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Premium supplier since 2011 | ||||
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Classification | Chemical reagent >> Organic reagent >> Ester >> Lactone compound |
|---|---|
| Name | 1,3-Propanesultone |
| Synonyms | 1,2-Oxathiolane 2,2-dioxide; 3-Hydroxy-1-propanesulfonic acid gamma-sultone; Propane sultone |
| Molecular Structure | 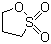 |
| Molecular Formula | C3H6O3S |
| Molecular Weight | 122.14 |
| CAS Registry Number | 1120-71-4 |
| EC Number | 214-317-9 |
| SMILES | C1COS(=O)(=O)C1 |
| Water solubility | Slightly soluble 10.0 g/100g (Expl.) |
|---|---|
| Density | 1.4±0.1 g/cm3, Calc.*, 1.392 g/mL (Expl.) |
| Melting point | 30-33 °C (Expl.) |
| Index of Refraction | 1.478, Calc.* |
| Boiling Point | 297.6±9.0 °C (760 mmHg), Calc.*,180 °C (30 mmHg) (Expl.) |
| Flash Point | 133.8±18.7 °C, Calc.*, 113 °C (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS08;GHS07 Danger Details GHS08;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H350-H312-H302 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P261-P262-P264-P264+P265-P270-P271-P273-P280-P301+P316-P301+P317-P302+P352-P304+P340-P305+P351+P338-P305+P354+P338-P316-P317-P318-P321-P330-P332+P317-P337+P317-P361+P364-P362+P364-P405- Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2810 | ||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||
|
1,3-Propanesultone is an organic compound that belongs to the class of sulfonates, which is characterized by a three-membered sulfonate ring. It is commonly used as a reagent in organic synthesis due to its electrophilic properties, particularly its ability to react with nucleophilic compounds. The compound’s structure consists of a sulfonate group (-SO3) attached to a three-carbon alkyl chain, making it both a useful intermediate in chemical reactions and a versatile building block in the production of a variety of chemicals. The discovery of 1,3-propanesultone dates back to the mid-20th century, when chemists began to explore the reactivity of sulfonates in organic chemistry. Sulfonates were known for their ability to undergo nucleophilic substitution reactions, which led to their use in the synthesis of a wide range of compounds, including pharmaceuticals, agricultural chemicals, and industrial materials. The unique properties of 1,3-propanesultone, such as its ability to undergo ring-opening reactions, were quickly recognized, making it an important compound in both industrial and academic research. One of the primary applications of 1,3-propanesultone is in the synthesis of sulfonate-based surfactants. Its high reactivity allows it to react with alcohols and amines to form a variety of functionalized derivatives. These derivatives are often used as intermediates in the production of detergents, emulsifiers, and other surface-active agents. Due to its ability to modify surface properties, 1,3-propanesultone and its derivatives are used in the formulation of cleaning products, coatings, and materials designed for improved wettability and spreading properties. In addition to its use in the production of surfactants, 1,3-propanesultone has important applications in the field of polymer chemistry. It serves as a cross-linking agent in the preparation of high-performance polymers, particularly in the production of polyurethanes and epoxy resins. By reacting with diols or amines, 1,3-propanesultone can form networks of cross-linked polymer chains, which impart increased mechanical strength, thermal stability, and chemical resistance to the resulting materials. These properties make 1,3-propanesultone-containing polymers suitable for use in coatings, adhesives, and other industrial applications that require robust performance. 1,3-Propanesultone is also used in the preparation of various heterocyclic compounds through nucleophilic substitution reactions. The sulfonate group of 1,3-propanesultone is highly reactive toward nucleophilic attack, enabling its use in the synthesis of sulfonated heterocycles, which are important intermediates in the production of pharmaceuticals, agrochemicals, and fine chemicals. By modifying the structure of 1,3-propanesultone, it is possible to create compounds with enhanced biological activity, making it valuable in drug discovery and the development of therapeutic agents. Another significant application of 1,3-propanesultone is in the field of battery technology. It has been explored as a potential electrolyte additive in lithium-ion batteries. The incorporation of 1,3-propanesultone into electrolyte solutions can enhance the stability and performance of batteries, particularly by improving the overall cycle life and charge-discharge efficiency. The compound’s ability to form stable complexes with metal ions makes it an ideal candidate for improving the efficiency and longevity of energy storage devices. In conclusion, 1,3-propanesultone is a highly versatile chemical with a range of applications in organic synthesis, polymer chemistry, and industrial materials. Its reactivity and ability to form functional derivatives make it an essential building block in the production of surfactants, polymers, and pharmaceuticals. Its use in energy storage systems further highlights its potential for innovation in modern technologies. References 2023. Review on low-temperature electrolytes for lithium-ion and lithium metal batteries. Electrochemical Energy Reviews. DOI: 10.1007/s41918-023-00199-1 2024. Bacterial cellulose/zwitterionic dual-network porous gel polymer electrolytes with high ionic conductivity. Journal of Wuhan University of Technology-Mater. Sci. Ed.. DOI: 10.1007/s11595-024-2915-5 2024. Investigation of the electrochemical conversion of brighteners during nickel electrodeposition, Part 1: chemical analysis by mass spectrometry. Journal of Applied Electrochemistry. DOI: 10.1007/s10800-024-02208-7 |
| Market Analysis Reports |