Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Ring Specialty Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.ringchemicals.com | |||
 | +1 (416) 493-6870 | |||
 | info@ringchemicals.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shandong Boyuan Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.boyuanpharm.com | |||
 | +86 (531) 6995-4981 8896-3280 +86 15806417970 | |||
 | +86 (531) 8896-4879 | |||
 | Jeffrey.Liu@boyuanpharm.com boyuanchem@126.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Ich Biofarm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ichemie.com | |||
 | +86 (571) 2818-6870 | |||
 | +86 (571) 8993-9479 | |||
 | specialchem1@ichemie.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Watec Laboratories, Inc. | China | |||
|---|---|---|---|---|
 | www.wateclaboratories.com | |||
 | +86 (519) 8692-1516 +86 18602586511 | |||
 | +86 (519) 8526-7382 | |||
 | info@wateclaboratories.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Kingreat Chemistry Company Limited | China | |||
|---|---|---|---|---|
 | www.chemtrade.cn | |||
 | +86 (592) 229-9609 | |||
 | +86 (592) 229-9601 | |||
 | kingreat@vip.163.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Synthesis With Catalysts Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.synthesiswithcatalysts.com | |||
 | +91 (120) 431-9102 | |||
 | +91 (120) 431-9102 | |||
 | sales1@synthesiswithcatalysts.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Vegomega Biopharma Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.veg-omega.com | |||
 | +86 18062552845 | |||
 | +86 (27) 8669-8696 | |||
 | vegomega@163.com | |||
| Chemical distributor since 2017 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Molekula Ltd | UK | |||
|---|---|---|---|---|
 | www.molekula.com | |||
 | +44 (1747) 831-066 | |||
 | +44 (1747) 831-199 | |||
 | info@molekula.com | |||
| Chemical manufacturer | ||||
| Arran Chemical Company | Ireland | |||
|---|---|---|---|---|
 | www.arranchemical.ie | |||
 | +353 (90) 644-5700 | |||
 | +353 (90) 649-4809 | |||
 | info@arranchemical.ie | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Polyamine |
|---|---|
| Name | (+/-)-trans-1,2-Diaminocyclohexane |
| Synonyms | (+/-)-trans-1,2-Cyclohexanediamine |
| Molecular Structure | 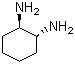 |
| Molecular Formula | C6H14N2 |
| Molecular Weight | 114.19 |
| CAS Registry Number | 1121-22-8 |
| EC Number | 601-163-6 |
| SMILES | C1CC[C@H]([C@@H](C1)N)N |
| Density | 0.951 g/mL |
|---|---|
| Melting point | 14-15 °C |
| Boiling point | 79-81 °C (15 mmHg) |
| Refractive index | 1.4885-1.4905 |
| Flash point | 68 °C |
| Water solubility | Soluble |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H314-H317 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P261-P264-P272-P280-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P354+P338-P316-P321-P333+P317-P362+P364-P363-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2735 | ||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
(+/-)-trans-1,2-Diaminocyclohexane is an organic compound consisting of a cyclohexane ring substituted with two amino (-NH2) groups at the 1 and 2 positions in a trans configuration. The term "(+/-)" indicates that the compound is a racemic mixture, containing equal amounts of both enantiomers (the (1R,2R) and (1S,2S) forms). Its molecular formula is C6H14N2. This compound is a key chiral diamine and has found broad utility as a ligand, intermediate, and building block in both academic research and industrial applications. The trans stereochemistry refers to the spatial arrangement of the two amino groups being on opposite sides of the cyclohexane ring, resulting in distinct chemical and physical properties compared to the cis isomer. The synthesis of racemic trans-1,2-diaminocyclohexane is typically achieved through hydrogenation of o-phenylenediamine or nitrocyclohexane derivatives, or via reductive amination processes involving cyclohexanedione intermediates. Alternatively, it can be derived from resolution of the cis isomer under specific catalytic or crystallization conditions. Commercially, the racemic trans form is widely available and cost-effective. One of the most important applications of (+/-)-trans-1,2-diaminocyclohexane is in coordination chemistry. It serves as a bidentate ligand, coordinating metal ions through both nitrogen atoms. Complexes formed with transition metals, such as platinum, ruthenium, and rhodium, are extensively used in catalysis, including asymmetric hydrogenation, oxidation, and C-C bond-forming reactions. Metal complexes of the enantiopure forms of this diamine are also used as chiral auxiliaries or catalysts in enantioselective synthesis. Another critical application is in the field of pharmaceuticals. While the racemate itself is not typically used as a drug, its derivatives and metal complexes have shown significant biological activity. For example, certain platinum complexes containing trans-1,2-diaminocyclohexane ligands exhibit antitumor activity and are structurally related to oxaliplatin, a clinically used anticancer drug. In polymer and material science, (+/-)-trans-1,2-diaminocyclohexane is used as a monomer or curing agent in the production of polyureas and polyimides, where its rigidity and stereochemistry contribute to thermal and mechanical stability of the resulting materials. Physically, the compound is a colorless to pale yellow solid at room temperature. It is soluble in water, alcohols, and many organic solvents. The two amino groups contribute to its basicity and reactivity, allowing it to participate in condensation, alkylation, acylation, and other transformations. As a primary diamine, it can also undergo reactions with aldehydes and ketones to form Schiff bases or with carboxylic acids and acid chlorides to form diamides. In handling, it should be noted that the compound is corrosive and may cause irritation to skin, eyes, and respiratory system. Proper protective equipment and ventilation are necessary during use. In summary, (+/-)-trans-1,2-diaminocyclohexane is a versatile, racemic chiral diamine widely employed in coordination chemistry, asymmetric catalysis, and materials synthesis. Its structural rigidity, trans stereochemistry, and dual amine functionality make it a valuable intermediate and ligand across diverse chemical disciplines. References 2018. Recent Developments in the Synthesis of Pyrido[1,2-a]benzimidazoles. Synthesis, 50(8). DOI: 10.1055/s-0036-1589533 2018. Asymmetric Synthesis of Adamantyl GABA Analogues. Synthesis, 50(19). DOI: 10.1055/s-0037-1610824 2024. Rational design of dual Chemo- and Thermo-responsive phase separation of Poly(4-hydroxystyrene) in non-aqueous media. Polymer Journal, 56(10). DOI: 10.1038/s41428-024-00968-9 |
| Market Analysis Reports |