Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Ring Specialty Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.ringchemicals.com | |||
 | +1 (416) 493-6870 | |||
 | info@ringchemicals.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2010 | ||||
| Watec Laboratories, Inc. | China | |||
|---|---|---|---|---|
 | www.wateclaboratories.com | |||
 | +86 (519) 8692-1516 +86 18602586511 | |||
 | +86 (519) 8526-7382 | |||
 | info@wateclaboratories.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Taizhou Tianhong Biochemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.thbiochem.com | |||
 | +86 (523) 8620-1489 | |||
 | +86 (523) 8613-2544 | |||
 | thbiochem@126.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Arran Chemical Company | Ireland | |||
|---|---|---|---|---|
 | www.arranchemical.ie | |||
 | +353 (90) 644-5700 | |||
 | +353 (90) 649-4809 | |||
 | info@arranchemical.ie | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| AK Scientific, Inc | USA | |||
|---|---|---|---|---|
 | www.aksci.com | |||
 | +1 (510) 429-8835 | |||
 | +1 (510) 429-8836 | |||
 | sales@aksci.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Imide |
|---|---|
| Name | cis-1,2-Diaminocyclohexane |
| Molecular Structure | 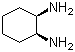 |
| Molecular Formula | C6H14N2 |
| Molecular Weight | 114.19 |
| CAS Registry Number | 1436-59-5 |
| EC Number | 604-372-0 |
| SMILES | C1CC[C@@H]([C@@H](C1)N)N |
| Density | 0.9$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 193.6 $degree$C 760 mmHg (Calc.)*, 220.9 - 222.2 $degree$C (Expl.) |
| Flash point | 75.0 $degree$C (Calc.)*, 70 $degree$C (Expl.) |
| Index of refraction | 1.484 (Calc.)*, 1.493 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS05 Danger Details GHS05 Danger Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H314 Details | ||||||||||||||||||||
| Safety Statements | P260-P264-P280-P301+P330+P331-P302+P361+P354-P304+P340-P305+P354+P338-P316-P321-P363-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| Transport Information | UN 2735 | ||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
cis-1,2-Diaminocyclohexane is an alicyclic diamine with the molecular formula C6H14N2. It consists of a cyclohexane ring bearing two amino groups (–NH2) attached to adjacent carbon atoms in a cis stereochemical configuration, meaning both amino substituents are oriented on the same side of the cyclohexane ring plane. The molecule belongs to the class of vicinal diamines, compounds containing two amino groups on neighboring carbon atoms. The cyclohexane ring adopts conformations similar to other saturated six-membered rings, primarily the chair conformation. In the cis isomer, one amino group occupies an axial position and the other an equatorial position in the most stable chair conformer. Ring flipping can interchange these positions while preserving the cis relationship between the substituents. The stereochemistry of cis-1,2-diaminocyclohexane is important because it strongly influences the compound’s coordination behavior, conformational properties, and reactivity. The molecule contains two stereogenic carbon centers, and the cis form exists as a pair of enantiomers. These stereochemical features make the compound valuable in asymmetric synthesis and coordination chemistry. The amino groups are basic and nucleophilic, allowing the compound to react readily with acids, aldehydes, ketones, acylating agents, and metal ions. Protonation of the amino groups yields diammonium salts, while condensation reactions with carbonyl compounds can form imines or Schiff bases. The close spatial arrangement of the two amino groups also enables bidentate coordination to metal centers. cis-1,2-Diaminocyclohexane has been widely used as a ligand and intermediate in coordination chemistry and organic synthesis. In metal complexes, the two nitrogen donor atoms can chelate to a single metal center, forming stable five-membered chelate rings. The stereochemistry of the ligand can influence the geometry and chirality of the resulting coordination complexes. The compound is also important in the synthesis of chelating agents and pharmaceutical intermediates. Derivatives of cyclohexanediamines have been used in the preparation of ligands, catalysts, and biologically active compounds. The rigid cyclohexane framework provides conformational control that is useful in stereoselective chemistry. Industrial and laboratory synthesis of cis-1,2-diaminocyclohexane typically involves reduction or hydrogenation of corresponding dinitro or diamide precursors, or stereoselective functionalization of cyclohexene derivatives. Separation of cis and trans isomers may be required because both stereoisomers are commonly formed in synthetic processes. From a physicochemical standpoint, cis-1,2-diaminocyclohexane is a strongly basic compound due to the presence of two primary amino groups. It is highly polar and capable of extensive hydrogen bonding. These properties contribute to its solubility behavior and reactivity in aqueous and polar organic media. Historically, cyclohexanediamine isomers gained importance through their applications in coordination chemistry and polymer science. The stereochemical differences between cis and trans isomers were found to significantly affect ligand geometry and complex stability, leading to extensive study of their conformational and coordination properties. Overall, cis-1,2-diaminocyclohexane is a stereochemically defined vicinal diamine featuring a cyclohexane ring with adjacent amino substituents on the same side of the ring. Its combination of conformational rigidity, bidentate coordination ability, and stereochemical control has made it an important compound in coordination chemistry, synthetic chemistry, and ligand design. References 2024. A charge-neutral organic cage selectively binds strongly hydrated sulfate anions in water. Nature Chemistry. DOI: 10.1038/s41557-024-01457-5 2023. Geometric-algebraic approach to aqueous solutions of diprotic acids and its buffer mixtures. Journal of Mathematical Chemistry. DOI: 10.1007/s10910-023-01527-y |
| Market Analysis Reports |