Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Minakem S.A.S. | France | |||
|---|---|---|---|---|
 | www.minakem.com | |||
 | +33 (3) 2064-6830 | |||
 | +33 (3) 2064-3198 | |||
 | contact@minakem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| 2A Pharmachem USA | USA | |||
|---|---|---|---|---|
 | www.2apharmachem.com | |||
 | +1 (630) 322-8887 | |||
 | +1 (630) 322-8885 | |||
 | sales@2abiotech.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shandong Boyuan Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.boyuanpharm.com | |||
 | +86 (531) 6995-4981 8896-3280 +86 15806417970 | |||
 | +86 (531) 8896-4879 | |||
 | Jeffrey.Liu@boyuanpharm.com boyuanchem@126.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Ich Biofarm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ichemie.com | |||
 | +86 (571) 2818-6870 | |||
 | +86 (571) 8993-9479 | |||
 | specialchem1@ichemie.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2011 | ||||
| NetQem, LLC | USA | |||
|---|---|---|---|---|
 | www.netqem.us | |||
 | +1 (919) 544-4122 | |||
 | +1 (919) 544-4109 | |||
 | sales@netqem.us | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Synthesis With Catalysts Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.synthesiswithcatalysts.com | |||
 | +91 (120) 431-9102 | |||
 | +91 (120) 431-9102 | |||
 | sales1@synthesiswithcatalysts.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Enantia, S.L. | Spain | |||
|---|---|---|---|---|
 | www.enantia.com | |||
 | +34 (93) 403-4837 | |||
 | +34 (93) 403-4838 | |||
 | info@enantia.com | |||
| Chemical manufacturer | ||||
| Arran Chemical Company | Ireland | |||
|---|---|---|---|---|
 | www.arranchemical.ie | |||
 | +353 (90) 644-5700 | |||
 | +353 (90) 649-4809 | |||
 | info@arranchemical.ie | |||
| Chemical manufacturer | ||||
| Acros Organics | Belgium | |||
|---|---|---|---|---|
 | www.acros.com | |||
 | +86 (21) 5258-1100 | |||
 | +86 (21) 5258-0119 | |||
 | info@acros.com | |||
| Chemical manufacturer | ||||
| PCI Synthesis, Inc. | USA | |||
|---|---|---|---|---|
 | www.pcisynthesis.com | |||
 | +1 (978) 462-5555 463-4853 | |||
 | +1 (978) 465-2057 | |||
 | sales@pcisynthesis.com | |||
| Chemical manufacturer | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| CNH Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.cnhtechnologies.com | |||
 | +1 (781) 933-0362 | |||
 | +1 (781) 933-1839 | |||
 | info@cnhtechnologies.com | |||
| Chemical manufacturer | ||||
| Toray Fine Chemicals Co., Ltd. | Japan | |||
|---|---|---|---|---|
 | www.torayfinechemicals.com | |||
 | +81 (47) 350-6219 | |||
 | +81 (47) 350-6091 | |||
 | info@torayfinechemicals.com | |||
| Chemical manufacturer | ||||
| Reuter Chemische Apparatebau KG | Germany | |||
|---|---|---|---|---|
 | www.rca-separations.de | |||
 | +49 (761) 559-6460 | |||
 | +49 (761) 559-6488 | |||
 | sales@rca-separations.de | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Polyamine |
|---|---|
| Name | (1R,2R)-(-)-1,2-Diaminocyclohexane |
| Synonyms | (R,R)-DACH |
| Molecular Structure | 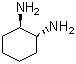 |
| Molecular Formula | C6H14N2 |
| Molecular Weight | 114.19 |
| CAS Registry Number | 20439-47-8 |
| EC Number | 606-556-6 |
| SMILES | C1CC[C@H]([C@@H](C1)N)N |
| Density | 0.9±0.1 g/cm3 Calc.*, 0.954 g/mL (Expl.) |
|---|---|
| Melting point | 40-45 °C (Expl.) |
| Boiling point | 193.6 °C 760 mmHg (Calc.)*, 220.9 - 222.2 °C (Expl.) |
| Flash point | 75.0 °C (Calc.)*, 70 °C (Expl.) |
| Index of refraction | 1.484 (Calc.)*, 1.49 (Expl.) |
| Alpha | -24.5 ° (c=5, 1 n hcl) (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H314-H317 Details | ||||||||||||||||||||
| Safety Statements | P260-P261-P264-P272-P280-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P354+P338-P316-P321-P333+P317-P362+P364-P363-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| Transport Information | UN 3259 | ||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
(1R,2R)-(−)-1,2-Diaminocyclohexane is the enantiomerically pure form of 1,2-diaminocyclohexane, featuring two amino groups positioned on adjacent carbons of a cyclohexane ring in a cis configuration. The (1R,2R) stereochemistry denotes the absolute configuration of the two chiral centers, and the (−) sign indicates that this is the levorotatory enantiomer, meaning it rotates plane-polarized light counterclockwise. This compound is widely used as a chiral ligand and building block in asymmetric synthesis due to its bidentate coordination ability through the two amino groups. The cis arrangement allows it to chelate metal centers effectively, making it valuable in the preparation of metal complexes that serve as catalysts in enantioselective transformations such as hydrogenation, allylic substitution, and cyclopropanation. The synthesis of enantiomerically pure (1R,2R)-1,2-diaminocyclohexane can be accomplished by resolution of the racemic mixture through diastereomeric salt formation or by asymmetric synthesis methods starting from chiral precursors. Its availability in optically pure form is crucial for applications requiring high enantioselectivity. In coordination chemistry, (1R,2R)-(−)-1,2-diaminocyclohexane forms stable chelate complexes with transition metals like platinum, rhodium, and ruthenium. These complexes are extensively studied and employed as catalysts in pharmaceutical and fine chemical manufacturing. The rigidity of the cyclohexane ring combined with the stereochemical arrangement provides well-defined chiral environments for catalytic reactions. Apart from catalysis, derivatives of this diamine are also investigated in medicinal chemistry for their potential biological activities. Metal complexes bearing this ligand have shown promise as antitumor agents and in other therapeutic contexts. Physically, (1R,2R)-(−)-1,2-diaminocyclohexane is a colorless to pale yellow liquid or crystalline solid, depending on purity and conditions. It is soluble in water and polar organic solvents due to the presence of two primary amine groups, which also contribute to its basicity and reactivity. Overall, (1R,2R)-(−)-1,2-diaminocyclohexane is a valuable chiral diamine with significant roles in asymmetric catalysis, coordination chemistry, and potential biomedical applications, owing to its defined stereochemistry and coordination capabilities. References 2007. Cyclophosphamide "metronomic" chemotherapy for palliative treatment of a young patient with advanced epithelial ovarian cancer. BMC Cancer, 7. DOI: 10.1186/1471-2407-7-65 2023. The synthesis of bi(poly)- and macrocyclic derivatives of trans-diaminocyclohexane (microreview). Chemistry of Heterocyclic Compounds, 59(11). DOI: 10.1007/s10593-023-03246-3 2023. Asymmetric organocatalysis: from a breakthrough methodology to sustainable catalysts and processes. Russian Chemical Bulletin, 72(1). DOI: 10.1007/s11172-023-3713-5 |
| Market Analysis Reports |