Online Database of Chemicals from Around the World
| Shanghai Oripharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.oripharm.com.cn | |||
 | +86 (21) 6439-6936 | |||
 | +86 (21) 6481-4386 | |||
 | info@oripharm.com.cn sales@oripharm.com.cn | |||
| Chemical distributor since 2003 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Shanghai ZaiQi Bio-Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemzq.com | |||
 | +86 (21) 5482-4098 | |||
 | +86 (21) 5482-4069 | |||
 | sales1@chemzq.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Classification | Biochemical >> Nucleoside drugs >> Nucleotides and their analogues |
|---|---|
| Name | 8-Azaadenine |
| Synonyms | 6-Amino-8-azapurine; 8-Aza-6-aminopurine; 1H-1,2,3-Triazolo[4,5-d]pyrimidin-7-amine; 7-Amino-1H-triazolo[4,5-d]pyrimidine |
| Molecular Structure | 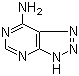 |
| Molecular Formula | C4H4N6 |
| Molecular Weight | 136.11 |
| CAS Registry Number | 1123-54-2 |
| EC Number | 214-375-5 |
| SMILES | C1=NC2=NNN=C2C(=N1)N |
| Density | 1.8±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 320 °C (Expl.) |
| Boiling point | 563.9±30.0 °C 760 mmHg (Calc.)* |
| Flash point | 329.4±11.8 °C (Calc.)* |
| Index of refraction | 1.875 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
8-Azaadenine (CAS 1123-54-2) is a purine base analogue (an “azapurine”) in which a nitrogen atom replaces the carbon at the 8-position of adenine. Its molecular formula is C4H4N8. Because of its structural similarity to adenine, it can interfere with purine metabolism, nucleotide biosynthesis, and nucleic acid function. The compound was characterized early as a purine metabolic antagonist. One of its significant biochemical effects is inhibition of xanthine oxidase, with reported 50 values in the sub-micromolar range, making it a useful tool in studies of purine catabolism. In cells, 8-azaadenine can be converted to corresponding nucleotide forms (e.g., 8-aza-AMP), which may act as “fraudulent” nucleotides, disrupting normal purine biosynthesis pathways. In medicinal chemistry, 8-azaadenine and related 8-azapurines have been used as scaffolds for the synthesis of biologically active derivatives. For instance, N6-substituted 8-azapurines have been synthesized and evaluated for antiplatelet activity. Structural modifications at the N6 position enabled tuning of biological potency and selectivity, leading to compounds with submicromolar activity in platelet aggregation assays. Beyond antiplatelet agents, 8-azaadenine derivatives have been explored in antiviral chemistry. Phosphonate analogues (such as 2-(phosphonomethoxy)alkyl derivatives) were prepared, though early studies noted that isomeric 8-aza derivatives (N7, N8, N9) showed varied activity. Some of these derivatives failed to exhibit strong antiviral activity against herpesviruses and HIV, underscoring the challenges of achieving therapeutic efficacy. On the structural and biophysical side, analogues of 8-azapurines have been incorporated into nucleosides and oligonucleotides. Modified nucleosides, such as 8-aza-7-deazaadenine, were synthesized and incorporated into DNA, where they displayed base-pairing capabilities with all four canonical nucleobases without major structural discrimination. These properties make 8-azapurines useful in nucleic acid research and in the design of novel nucleic acid-based probes or therapeutics. Because of its metabolic and biological activity, 8-azaadenine is also a prototypical mutagen in genetic and biochemical studies. Cells that lack functional adenine phosphoribosyltransferase (APRT) can resist its cytotoxicity because they are unable to convert it into lethal nucleotide forms, making it a tool for selecting APRT-deficient mutants. This property has been exploited in mutagenicity assays to study DNA repair, cell survival, and nucleotide pool regulation. Handling of 8-azaadenine in research requires consideration of its incorporation into nucleic acids and its metabolic conversion, which may generate toxic nucleotide analogues. In vitro and in vivo studies are conducted under controlled conditions to investigate its mechanism, metabolism, and downstream effects on cell cycle and genetic stability. Overall, 8-azaadenine is a well-established purine analogue that has contributed significantly to biochemical, genetic, and medicinal chemistry research. Its ability to perturb purine metabolism, interfere with nucleotide synthesis, and serve as a scaffold for biologically active derivatives makes it a versatile tool in both fundamental and applied science. References Zhao Z, Wang Y, Tian N, Yan H & Wang J (2021) Synthesis and biological evaluation of N6 derivatives of 8-azapurine as novel antiplatelet agents. RSC Medicinal Chemistry 12 1414–1427 DOI: 10.1039/D1MD00128K Seela F and Debelak H (2000) N8-(2′-deoxyribofuranosyl)-8-aza-7-deazaadenine: synthesis, base-pairing properties and incorporation into oligodeoxynucleotides. Nucleic Acids Research 28 17 3224–3232 DOI: 10.1093/nar/28.17.3224 |
| Market Analysis Reports |